Friday's health stories

Nebraska set to restrict abortion, gender-affirming care for trans minors
Nebraska lawmakers on Friday approved a bill to ban abortion at 12 weeks of pregnancy and restrict gender-affirming care for trans minors.
What's next: The bill, which is likely the first to combine restrictions on abortion and gender-affirming care, heads to the desk of Gov. Jim Pillen (R) for his signature.
FTC signals tougher rules for health companies' use of data
The Federal Trade Commission issued a warning Thursday about companies' use of consumers’ biometric information such as facial recognition technology, saying it raises "significant consumer privacy and data security concerns."
Why it matters: The warning comes as a growing number of companies amass data based on individuals' physiological features which could be used to infer consumers' health or other personal information.
What they're saying: "We view this policy statement as an important way to put companies on notice about the obligations they have under existing laws," Lina Khan said.
Catch up quick: The FTC has already been cracking down on the industry. For instance, on Thursday, the agency charged the developer of period tracking app Premom with deceiving users by sharing their health data with third parties, including Google and two China-based firms. Axios' Erin Brodwin wrote.
- But the nearly three-decade-old Health Insurance Portability and Accountability Act, or HIPAA, has grown less effective over time as the nature of information sharing has changed, Axios has previously reported.
- The FTC also proposed changes Thursday to the Health Breach Notification Rule, including modifying the definition of personal health record (PHR) identifiable health information and adding new definitions of health care provider and health care services.
- The throughline between PreMom, as well as GoodRx and Better Help "underscore how seriously we take protecting Americans' privacy, especially when it relates to people's most sensitive information," FTC chairwoman Lina Khan said.
- "Business models that are based on monetizing people's data can lead to situations where companies Americans are trusting with their sensitive data are then exposing that information for the sake of targeted advertising, analytics and engagement," Khan said.
Zoom in: It's not hard to imagine the possibility of a gaming VR headset collecting biometric data that indicates a kid's likelihood of developing dementia in older age, then selling that data to an insurance company, R.J. Cross, director of the Don't Sell My Data campaign at the organization U.S. PIRG, said during the public comment portion of an FTC meeting on Thursday.
- "Right now there are very few rules protecting our data," Cross told Axios, saying the FTC biometric policy statement is "a nice start" but will need to go bigger.
- "Every single one of us has data ricocheting around the databases of companies we’ve never heard of, used for all kinds of purposes we had no idea are happening," Cross said. "The problem only gets worse when you add sensitive physiological data into the mix."

Axios-Ipsos poll: "No clear agreement" on Americans' LGBTQ opinions
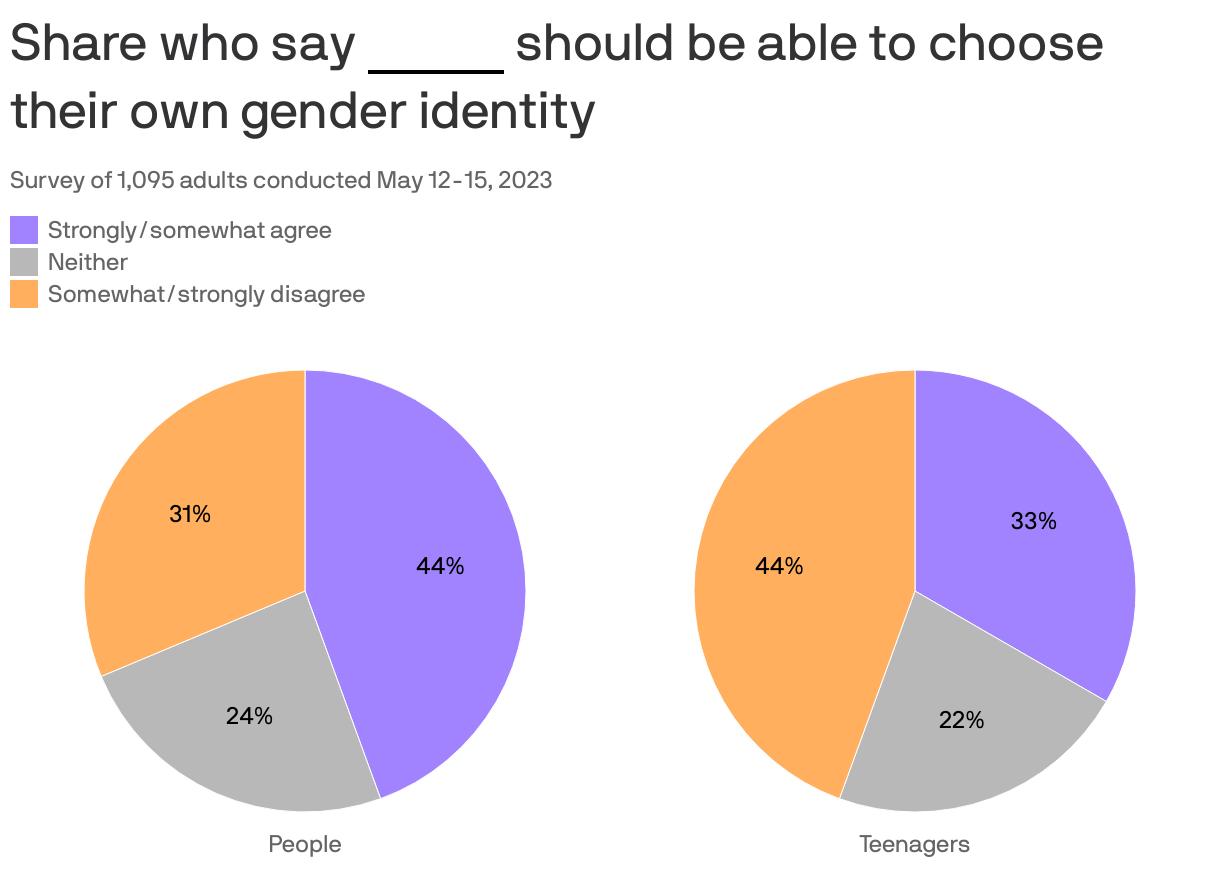
Many Americans lack fully developed views on LGBTQ issues and sexual identity as more states enact laws restricting access to transgender health care, according to the latest Axios-Ipsos American Health Index.
Why it matters: The views of a large segment of voters could be highly malleable as politicians elevate the issue in the run-up to the 2024 elections.
Hearing aids gain hipper reputation as "lifestyle" products after going OTC
Consumer electronics giants like Sony and Bose have staked out a piece of the new market for over-the-counter hearing aids, disrupting a $10 billion segment that's been dominated by niche audiology companies.
The big picture: An FDA rule finalized seven months ago created easier-to-access options for the 30 million people in the U.S. suffering from hearing loss — and is making devices that once carried a stigma more of modern lifestyle convenience.

House panel moves FDA funding bill with abortion and tobacco riders
The House Appropriations subcommittee in charge of FDA funding easily approved a fiscal 2024 spending bill Thursday that would reverse the agency's decision to allow mail-order mifepristone.
- The bill would also prevent HHS from banning menthol in cigarettes and from setting a maximum nicotine level in cigarettes.

Texas set to ban trans medical care for minors
A bill that would prohibit doctors from providing some gender-affirming care to children under 18 is headed to Texas Gov. Greg Abbott's desk after the legislation passed in the state's Republican-controlled Senate on Wednesday.
The big picture: The Senate voted 19-12 in favor of the bill that would bar transgender children from accessing surgeries, puberty blockers and hormone therapies on International Day Against Homophobia, Biphobia, and Transphobia.

FDA advisers endorse maternal vaccine for RSV
Food and Drug Administration advisers on Thursday endorsed giving pregnant people an experimental Pfizer vaccine to protect infants against respiratory syncytial virus, despite concerns about insufficient data on safety risks.
Why it matters: RSV is a leading cause of death for infants worldwide, particularly those under six months, but finding an effective vaccine has challenged scientists for decades.
How it works: Pfizer’s vaccine would prevent severe illness in infants in their first six months by creating neutralizing antibodies that can be transferred in the womb.
- Trial data analyzed by federal regulators found that the shot had an 82% efficacy rate at protecting infants in their first three months.
- It had a 69% efficacy rate at preventing severe illness at six months.
Yes, but: While the FDA’s 14 outside advisers unanimously voted the shot was effective, four voted no on the question of whether it was safe, saying there wasn't enough data.
- Some panelists noted concerns about preterm births — a safety risk which led GlaxoSmithKline to halt a trial of an RSV vaccine last year. A Pfizer official said there is no definitive evidence of increased risk.
- Other members, including Holly Janes, a biostatistician at Fred Hutchinson Cancer Center, said the stated optimal timing for administering the vaccine is still unclear.
- “I’m uncomfortable with the notion of kicking the can down the road” and relying on surveillance studies instead of trial data to confirm its safety, Janes said.
- Amanda Cohn, the CDC director of the division of birth defects and infant disorders, who voted yes for safety, worried about the FDA's ability to have a post-marketing surveillance system in place to track maternal health outcomes.
What they’re saying: Determining whether the benefits outweighs the risk is a “very complicated question because the person that we’re vaccinating is not the person who's receiving the benefit,” said Jay Portnoy, a professor of pediatrics at the University of Missouri-Kansas City.
- “I wish we had an explicit vote on maternal safety “ instead of one solely focused on the infant, said Saad Omer, an epidemiologist who leads the Yale Institute for Global Health. “Women are not just vessels.”
Catch up quick: Children’s hospitals last fall were swamped with respiratory infection cases.
- RSV treatment costs nearly $710 million per year, with public sources paying more for than half of infant RSV medical costs, according to an August analysis from the Journal of Infectious Diseases.
- The FDA earlier this month approved GlaxoSmithKline’s RSV vaccine for people 60 and older, another high-risk group.
- An FDA decision on Pfizer’s vaccine for the same age group is expected at the end of May.
Go deeper: Race for RSV shots heats up with maternal vaccine

FTC alleges Premom fertility app shared sensitive health data
The Federal Trade Commission has charged the developer of period tracking app Premom with deceiving users by sharing their health data with third parties, including Google and two China-based firms.
Why it matters: Legal protections for sensitive personal health data that can include information such as medications, mental health and pregnancy status have not kept up with the digital health sector's explosive growth.
Driving the news: The ruling is part of a concentrated recent effort by the FTC under activist chair Lina Khan to target a growing class of companies that are not subject to HIPAA yet nonetheless share and sell health data.
Details: According to the FTC, Premom shared users' reproductive health information and precise geolocation data with companies including Google and Alibaba subsidiary Umeng via tracking tools known as software development kits (SDKs).
- That data sharing "led to the unauthorized disclosure of facts about an individual user’s sexual and reproductive health, parental and pregnancy status," per the agency, as well as users' social media account data, precise geolocation data, and information about users' mobile devices and Wi-Fi network identifiers, the FTC says.
What's happening: The FTC is fining Premom $100,000 in civil penalties.
- If approved by the federal court, the order would bar Premom's parent company, Easy Healthcare Corporation, from sharing users' personal health data with third parties for advertising.
- It would also require Premom to get user consent before sharing such data.
What they're saying: "Premom broke its promises and compromised consumers’ privacy," Samuel Levine, director of the FTC’s Bureau of Consumer Protection, said in a statement.
Catch up quick: Wednesday's ruling follows similar actions against companies including period tracking app Flo, prescription discount startup GoodRx, and Teladoc mental health subsidiary BetterHelp.
Yes, and: Health privacy in the post-Roe digital age is fraught as prosecutors seeking to enforce anti-abortion laws are free to go after reproductive health data in mobile apps, where it is unprotected by federal law.
- As many as one-third of women use digital tools to track their periods, which can be for monitoring their cycles, planning to avoid a pregnancy or trying to conceive.
The bottom line: The FTC's moves directly challenge the way digital health companies operate and could significantly alter the future health tech landscape.
Kelly Tyko contributed to this story.

More than $1 billion in health insurance rebates projected to be paid this year
Insurers will have to rebate about $1.1 billion to enrollees this year for not spending enough of their premium revenues on medical claims under the Affordable Care Act, a KFF analysis shows.
Why it matters: Since rebates are based on a three-year average of insurers' experience, the givebacks reflects the pandemic experience, when patients canceled elective procedures and generally used less care.

These cities have the most bicycle deaths per capita
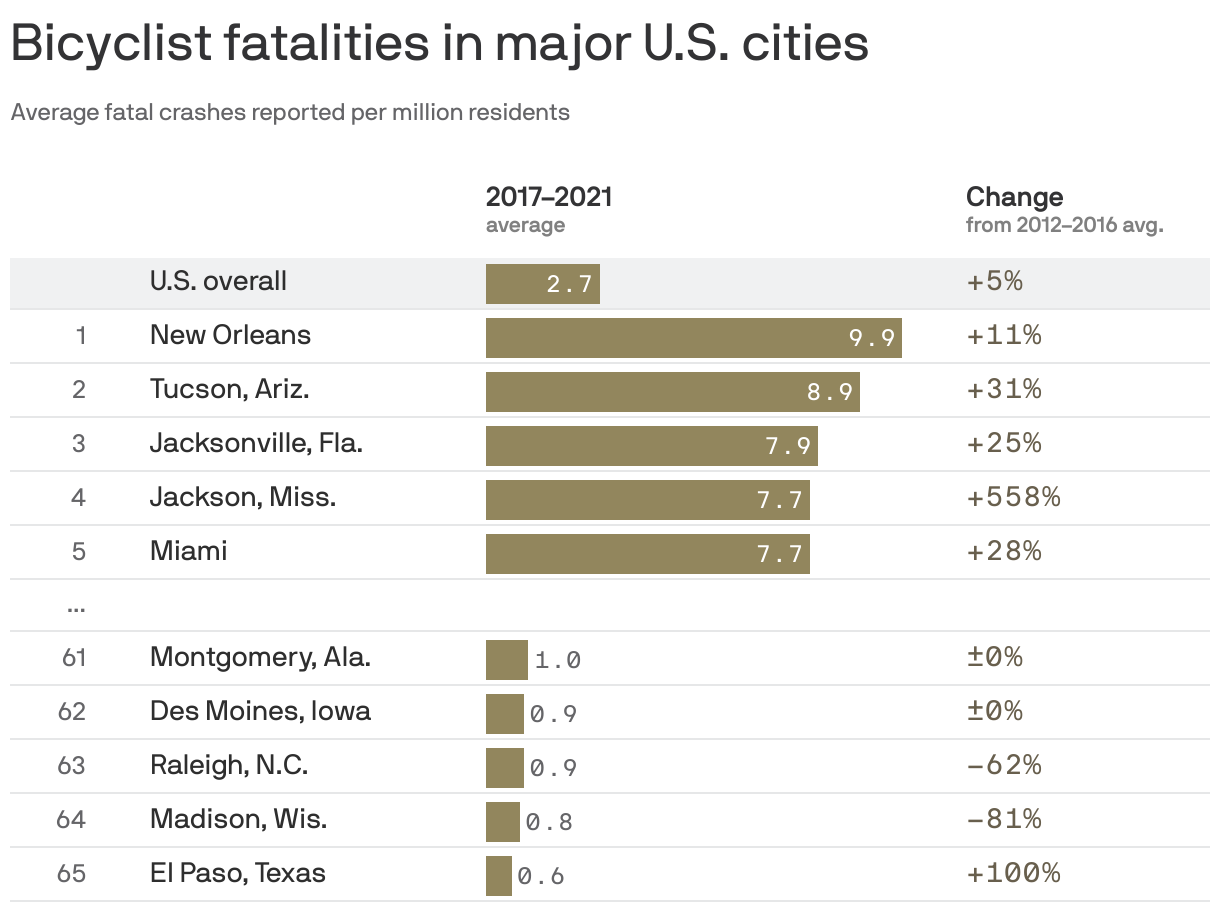
There were 2.7 fatal bicycle crashes on average for every million U.S. residents between 2017-2021, per data from the League of American Bicyclists via the National Highway Traffic Safety Administration — up 5% from 2012-2016.
- New Orleans (9.9), Tucson (8.9) and Jacksonville (7.9) had the country's highest rates of fatal accidents per million residents among major metro areas.

House panel advances transparency and PBM bills
A House Energy and Commerce Committee health markup on Wednesday offered more evidence that price transparency and pharmacy benefit manager regulation are two issues that have enough bipartisan support to move ahead in this Congress.
Among the measures the panel advanced on a unanimous 27-0 vote:

Axios-Ipsos poll: Gun violence soars as public health concern
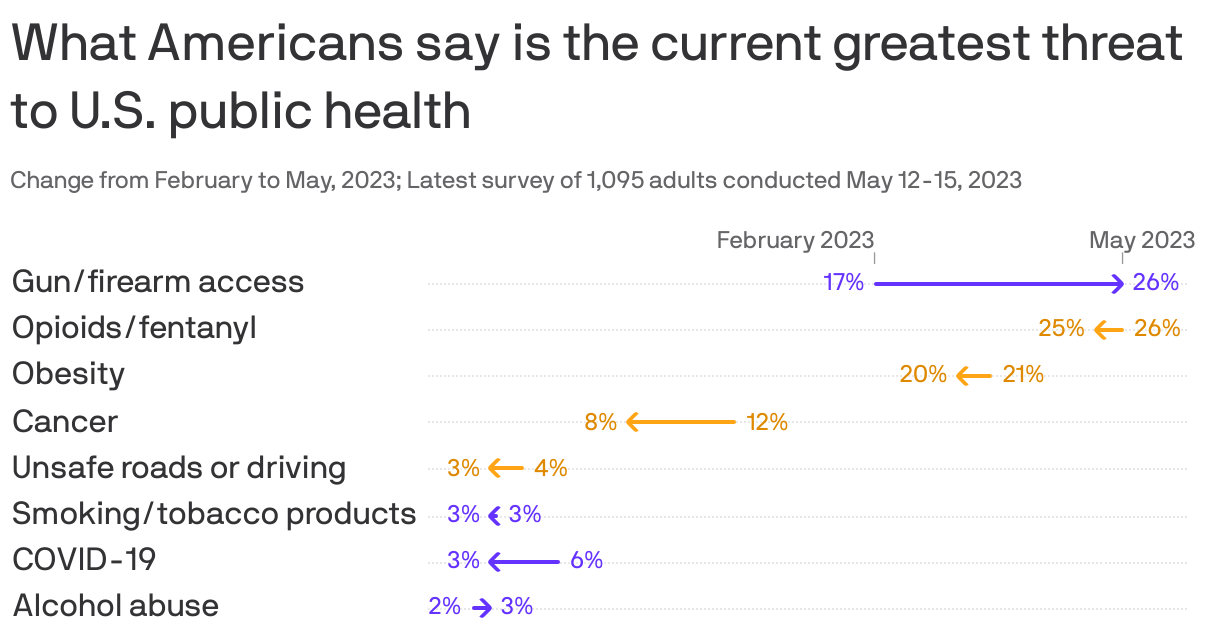
Gun violence has surged ahead of the opioid crisis as Americans' top public health concern, according to the latest edition of the Axios-Ipsos American Health Index.
Why it matters: A spate of recent mass shootings may have focused attention on the issue in ways that weren't apparent in our last poll in February.

Axios-Ipsos poll: Americans back work requirements for federal aid
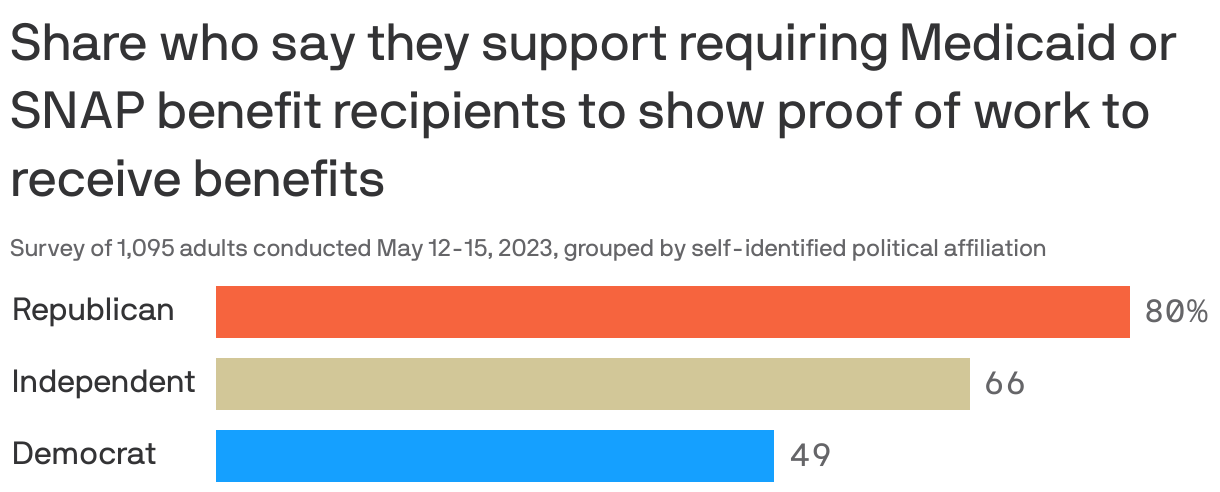
Nearly two-thirds of Americans — including half of Democrats — back work requirements for Medicaid and Supplemental Nutritional Assistance Program benefits, as House Republicans want in a debt limit deal, according to the latest Axios-Ipsos American Health Index.
Why it matters: Work requirements are a major sticking point in debt ceiling talks.

Theranos founder Elizabeth Holmes must start prison sentence May 30
Theranos founder Elizabeth Holmes must begin serving her prison sentence by May 30 while she appeals her conviction on charges of defrauding investors in her failed blood-testing startup, a federal judge ruled Wednesday.
Driving the news: U.S. District Court Judge Edward Davila issued the order after the United States Court of Appeals for the Ninth Circuit on Tuesday rejected Holmes' request to remain on bail as she appeals the case.