Monday's health stories

FDA approves Pfizer’s maternal RSV vaccine
The Food and Drug Administration on Monday approved the first vaccine that can be given to pregnant patients to protect infants against respiratory syncytial virus, or RSV.
Why it matters: The shot from Pfizer would give built-in protection against RSV to newborns, when they're most vulnerable to a virus that is a leading cause of death for infants worldwide and overwhelmed children's hospitals last fall.
Of note: Another new drug, Beyfortus, from Sanofi and AstraZeneca will be available for infants younger than 8 months old born during their first season of RSV, in addition to those between 8 and 19 months who are at high risk.
Details: Pfizer's vaccine, Abrysvo — which was previously approved for older adults — protects infants from severe RSV in their first six months by creating antibodies that neutralize the virus and can be transferred in the womb.
- Late-stage trial data found the vaccine was 82% effective at protecting infants from severe disease in their first three months and was 69% effective over six months.
- It will be administered as a single-dose injection between weeks 32 and 36 of pregnancy, per an FDA statement.
- This is a more limited window than what FDA advisers recommended in May, when they endorsed giving the shot as early as 24 weeks However, panel members said the data left the optimal timing unclear.
What they're saying: "This approval provides an option for healthcare providers and pregnant individuals to protect infants from this potentially life-threatening disease," said Peter Marks, director of FDA's Center for Biologics and Research.
Catch up fast: Independent advisers to the FDA in May endorsed Pfizer's vaccine to protect infants but expressed concerns about insufficient data on safety.
- Some panel members worried about the FDA's ability to track maternal health outcomes after the vaccine's approval.
- Other panelists noted the risk of preterm births, which led GlaxoSmithKline to halt a trial of an RSV vaccine last year.
- Pfizer officials said there is no definitive evidence of increased risk.
What's next: A CDC panel of outside vaccination experts still have to formally recommend the vaccine before it can be used in pregnant patients.
- The panel is scheduled to meet in October, but with RSV season approaching, it could call a special meeting before then.

Freeze of Georgia law adds twist to state efforts to ban trans care
A federal judge's decision to block part of Georgia's ban on most gender-affirming care for minors is throwing a new legal wrinkle into state efforts to restrict care for transgender youths.
Driving the news: U.S. District Court Judge Sarah Geraghty, a Biden appointee, wrote the state's hormone-therapy ban likely violates the Constitution's Equal Protection Clause and carries imminent risks of depression, anxiety, disordered eating, self-harm and suicidal ideation for transgender youths.

Gun deaths among U.S. children hit a new record high


Firearm deaths among children in the U.S. hit a new record high in 2021, according to a study published Monday in the journal Pediatrics.
Driving the news: The study, based on government data, points to the worsening of an already distressing trend, after guns became the leading cause of death for children for the first time in 2020.

This Kennedy scion is investing in disability-focused startups
A Kennedy family scion is raising a venture capital fund to invest in startups that serve people with disabilities, plus their caregivers and family members.
Why it matters: An estimated 1.3 billion people have a significant physical or mental disability, representing 16% of the global population, according to the World Health Organization,
Medical debt is squeezing millions of middle-class Americans, report says
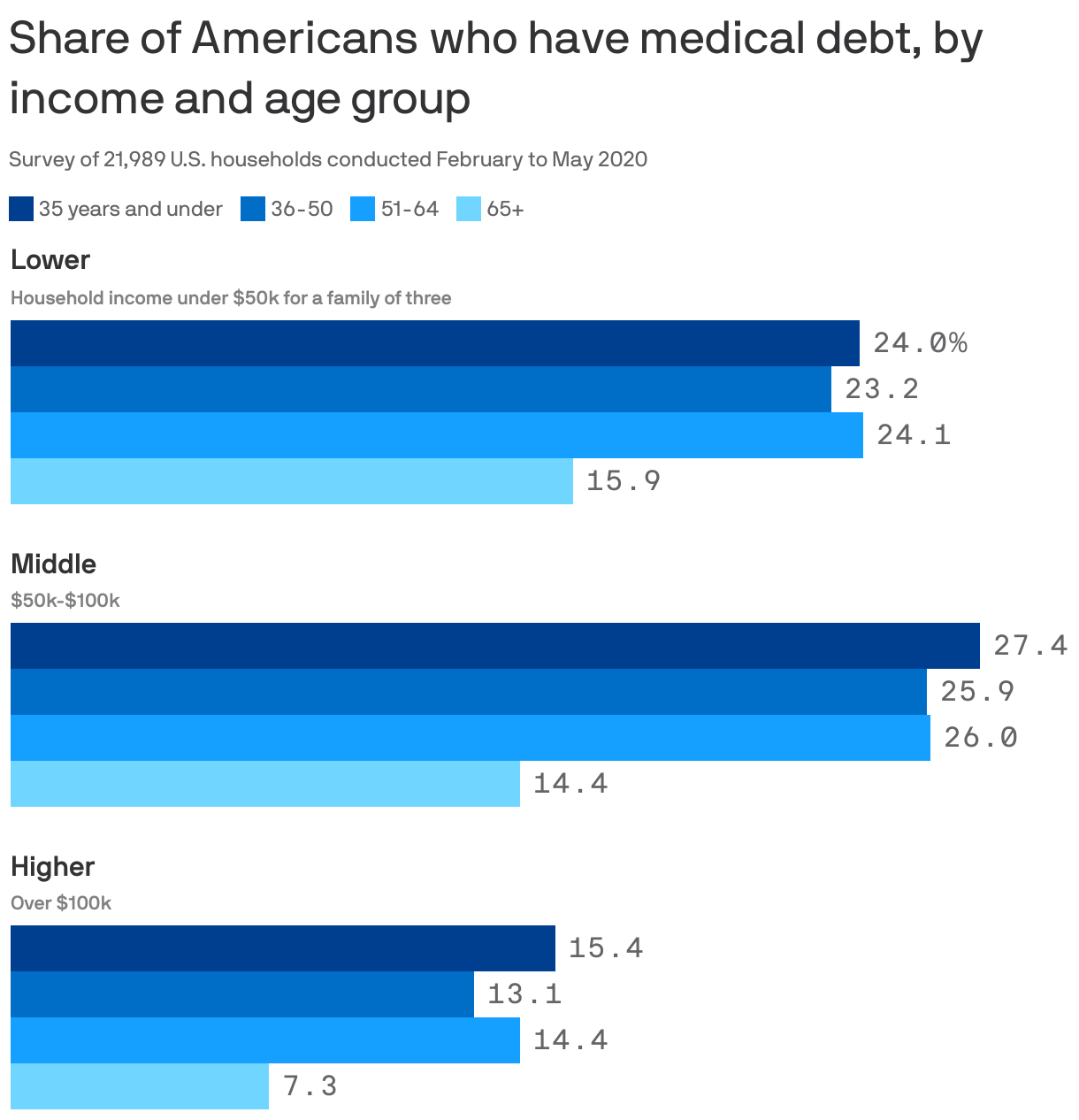
Middle-class Americans are the most likely to be saddled with medical debt, with nearly 1 in 4 — or roughly 17 million people — having unpaid medical bills, according to a report shared first with Axios from center-left think tank Third Way.
Driving the news: Middle-income Americans, who earn $50,000-$100,000 a year, are more likely than those with lower incomes to seek care but don't qualify for Medicaid or charity care to help pay for it.

Funding for cops stokes debate over use of opioid settlement money
As states and localities collect the first billions in opioid settlement funds, there's mounting concern over how some of the money is headed to law enforcement and plugging budget gaps instead of public health needs.
The big picture: At least 70% of the money has to be directed to opioid-related expenses, but critics fear limited transparency over the way it's being allocated risks crowding out the most pressing priorities and repeating the largely failed tactics of the war on drugs.

Rural hospitals feel squeeze from Medicare Advantage
Already struggling rural hospitals see an increasing financial threat from the steady growth in Medicare Advantage enrollment.
Why it matters: Lacking the bargaining powers of their larger peers who depend on commercial payers to turn a profit, some rural hospitals are losing money on private coverage like Medicare Advantage. The Medicare alternative's popularity with seniors is cutting into a typically better funding source for rural hospitals — traditional Medicare — as hundreds of rural hospitals face down financial calamity.