Seattle startup gets FDA clearance for new surgical imaging tool
Add Axios as your preferred source to
see more of our stories on Google.
Courtesy of Proprio
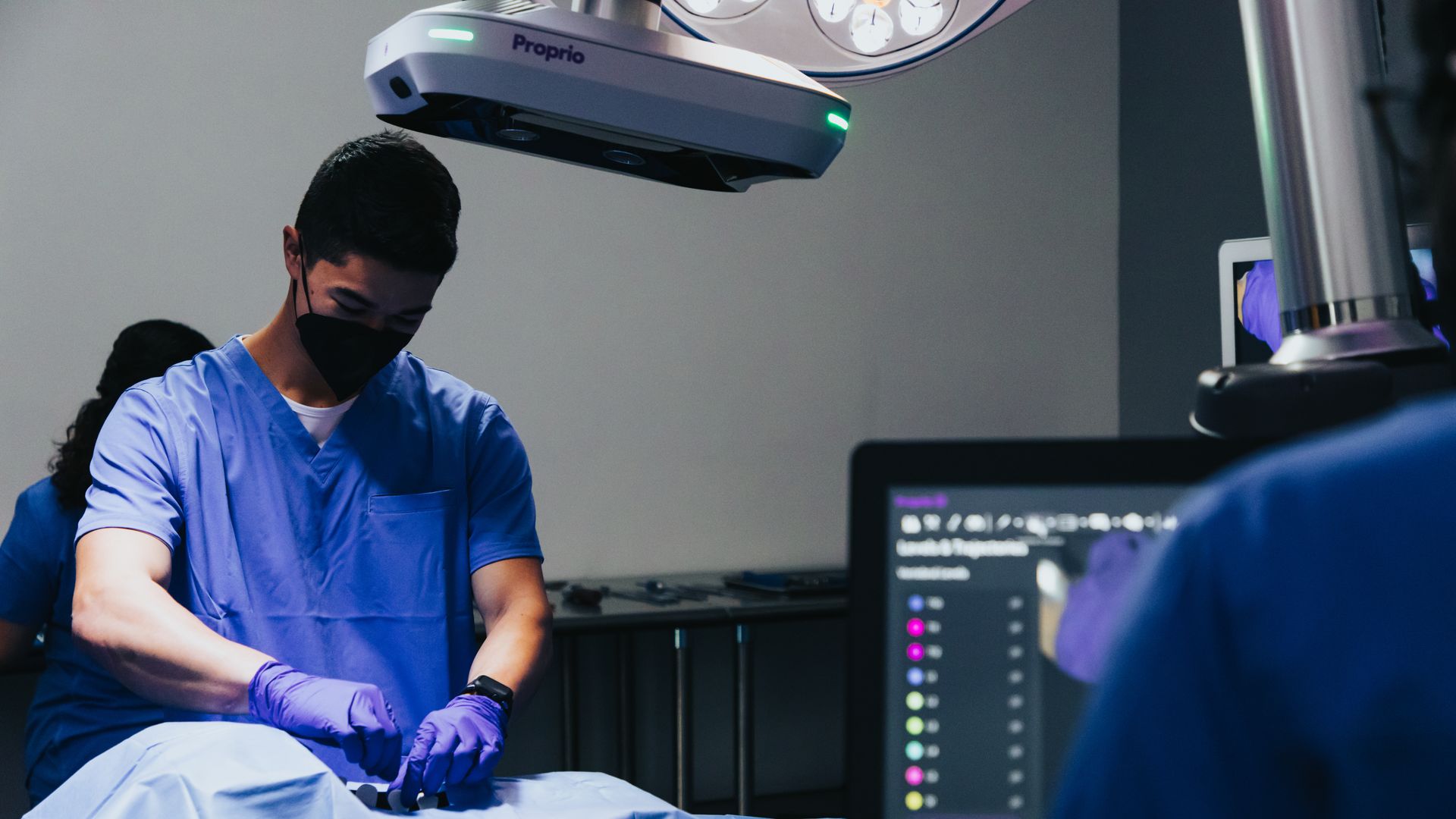
A Seattle medical tech company's revolutionary surgical imaging instrument has become the first light-enabled navigation tool for spine surgery to receive Food and Drug Administration clearance.
Why it matters: Officials with Proprio believe the tool, which allows surgeons to essentially "see" the spine being operated on in real time, could dramatically improve clinical outcomes.
Driving the news: This month, the Paradigm device received the FDA 510(k) clearance that's required for new medical implements before they are taken to market, Proprio said in an emailed announcement.
How it works: Using advancements in light field computer vision and AI, the system combines views of the spine from multiple sources to create a 3D visual image in real time.
- The system uses sensors to capture high-definition images and fuse that information with preoperative scans.
- Surgeons can therefore "see" the spine and surrounding anatomy during procedures, eliminating the need for mid-operation X-rays and scans.
- Removing the need for high-radiation intraoperative scans throughout the procedure can lead to a 10-time reduction in radiation exposure, Proprio officials said.
What they're saying: Samuel Browd, chief medical officer and co-founder of Proprio, told Axios that most of the spinal surgery navigation tools used today haven't been meaningfully updated in over 20 years, and the same goes for CT scans and X-rays.
- What used to require a time-consuming new image of the spine between every step of surgery can be replicated by the press of a button in under two seconds, he said.
- Browd said the old tools are "like using a printed atlas to navigate your route and driving straight into a traffic jam. Now, the surgical equivalent to Google Maps is available and will suggest you take an alternate route."
