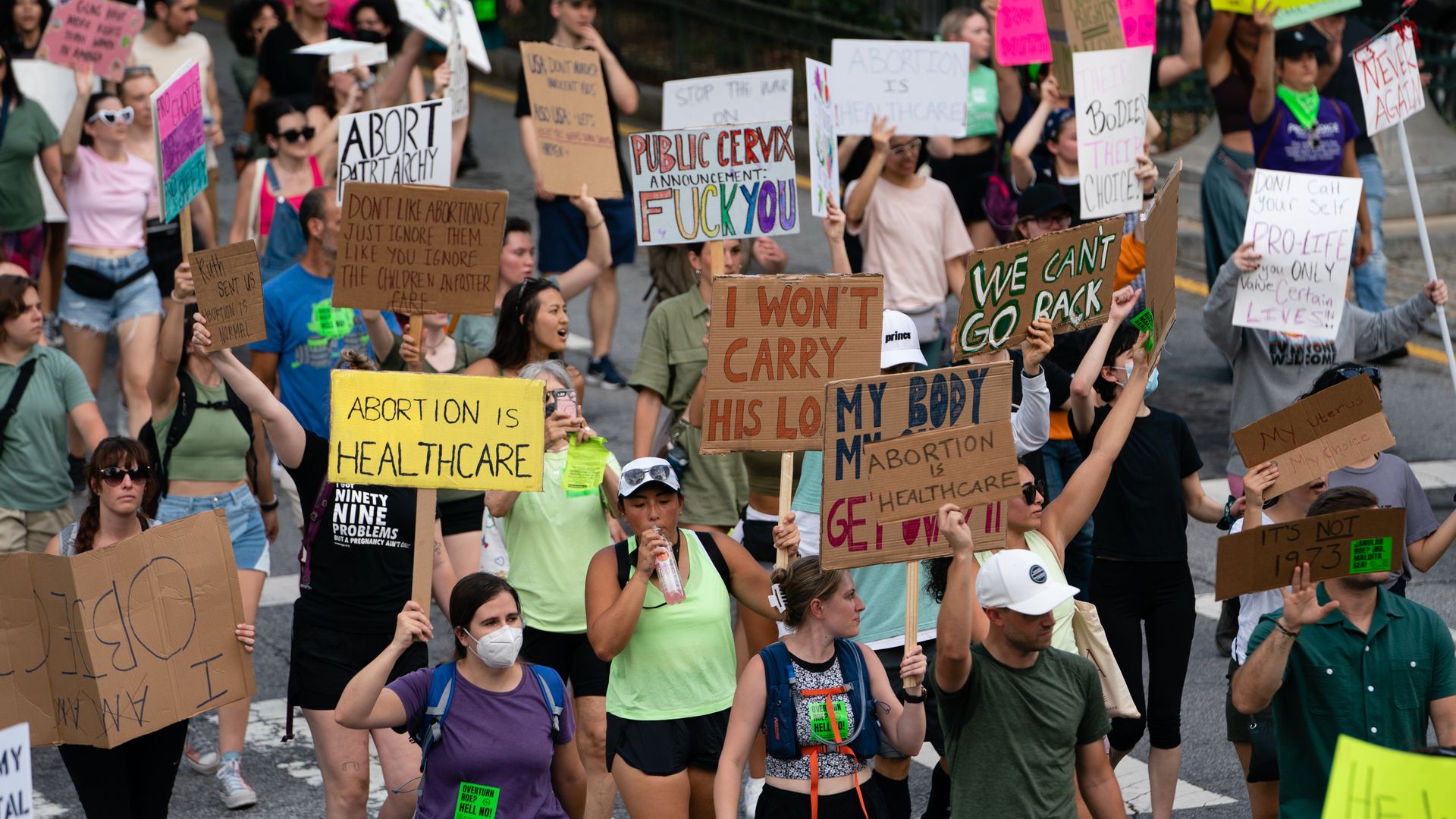Thursday's health stories

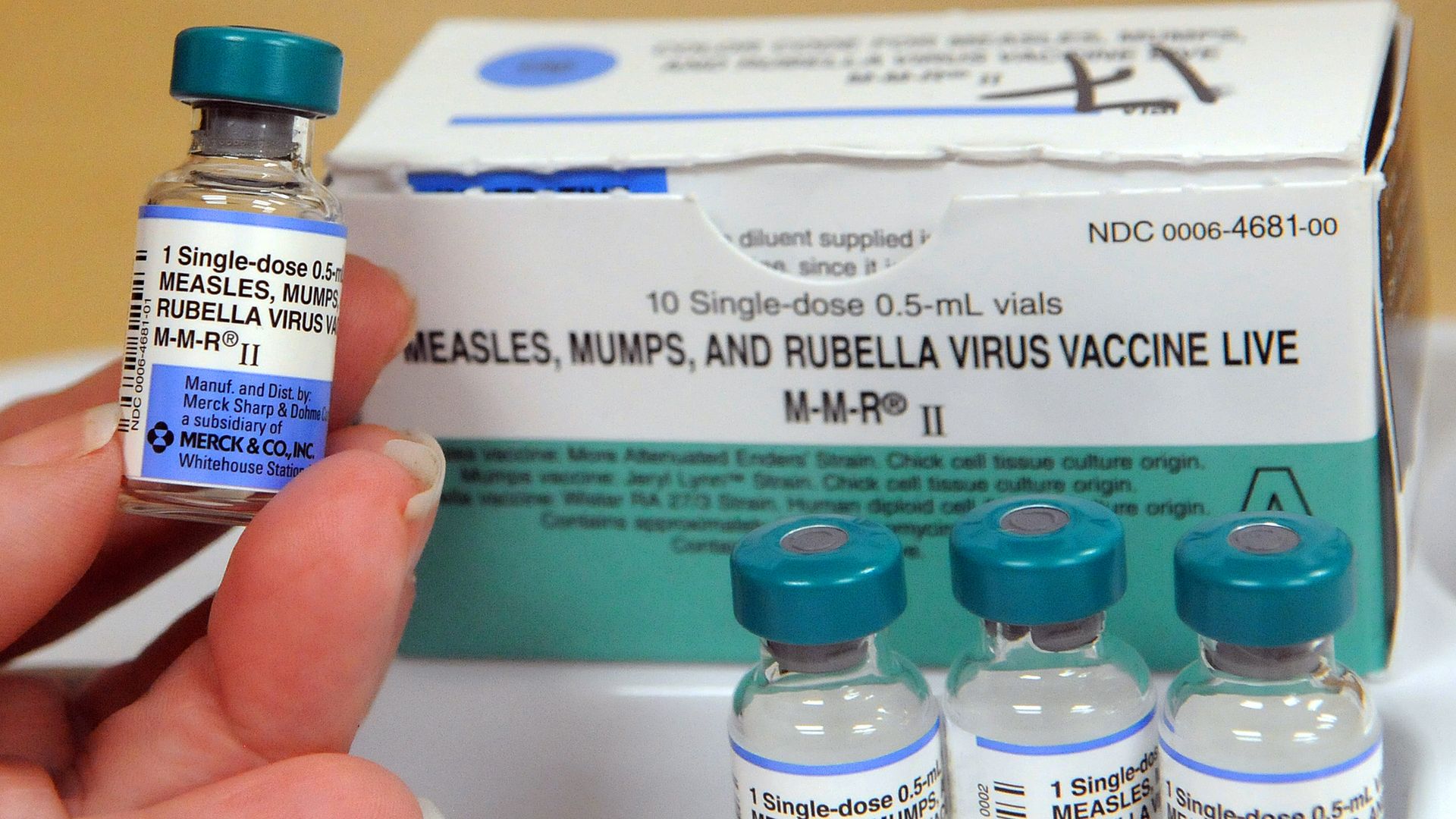
CDC and WHO say measles "an imminent threat" to world
Measles vaccination rates have steadily declined around the world since the start of the COVID-19 pandemic, leaving millions of children susceptible to the virus, according to new data in a joint report by the Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO).
Why it matters: The organizations said measles is "an imminent threat in every region of the world," as it is one of the most contagious human viruses — though it is also entirely preventable through vaccination.


Georgia Supreme Court reinstates state's 6-week abortion ban
The Georgia state Supreme Court on Wednesday temporarily reinstated the state's six-week abortion ban, which had been struck down by a lower court last week.
The big picture: The Georgia attorney general indicated they plan to appeal last week's decision on the ban shortly after it was issued. They simultaneously asked the state Supreme Court to temporarily “stay” the lower court ruling and reinstate the law in the meantime.

FDA approves gene therapy for hemophilia
The Food and Drug Administration on Tuesday approved a gene therapy for hemophilia — the latest in a series of decisions to advance pricey, personalized treatments that bring new hope to patients, along with cost concerns to the health system.
Why it matters: With a list price of $3.5 million, Hemgenix from CSL Behring LLC will become the most expensive therapy in the world.
- As Axios' Caitlin Owens reported in September, while the expected number of patients who'll receive such treatments is relatively low, employers, insurers, and public payers like state Medicaid programs may absorb large costs all at once.
- The Institute for Clinical and Economic Review this month suggested the highest price that should be charged for the treatment is about $2.9 million.
Details: Hemgenix is used to treat Hemophilia B, an inherited disorder affecting about one in 40,000 people that stems from missing or insufficient levels of a protein that's required to produce clots to stop bleeding.
- The treatment is given as a single dose by IV and contains a viral vector carrying a gene for the clotting factor.
- Its effectiveness was established based on decreases in the annualized bleeding rate of subjects in two studies.
- “Gene therapy for hemophilia has been on the horizon for more than two decades," said Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, adding Tuesday's decision "represents important progress in the development of innovative therapies for those experiencing a high burden of disease associated with this form of hemophilia.”
Catch up quick: The FDA recently approved two other multi-million-dollar gene therapies: Bluebird bio's Zynteglo for a blood disorder called beta-thalassemia and Skysona for a rare neurological disorder called cerebral adrenoleukodystrophy.
The growing menopause-at-work market
Menopause — a condition that's little discussed and poorly understood — is gaining more attention from employers rethinking the health benefits they offer women.
Why it matters: Support for workers experiencing telltale symptoms like hot flashes, fatigue and mood swings is becoming more essential after the pandemic and its economic shockwaves led a disproportionate number of women to exit the workforce.

Axios Finish Line: The power of active gratitude
This article originally appeared in Axios Finish Line, our nightly newsletter on life, leadership and wellness. Sign up here.
Whether we spend our time dwelling on things we're grateful for or noodling on things that upset us matters.
🖼️ The big picture: Shifting our attitudes to focus on giving thanks — not just during the holiday — has a direct link to our wellbeing.