Which age groups are eligible for the different COVID vaccines
Add Axios as your preferred source to
see more of our stories on Google.
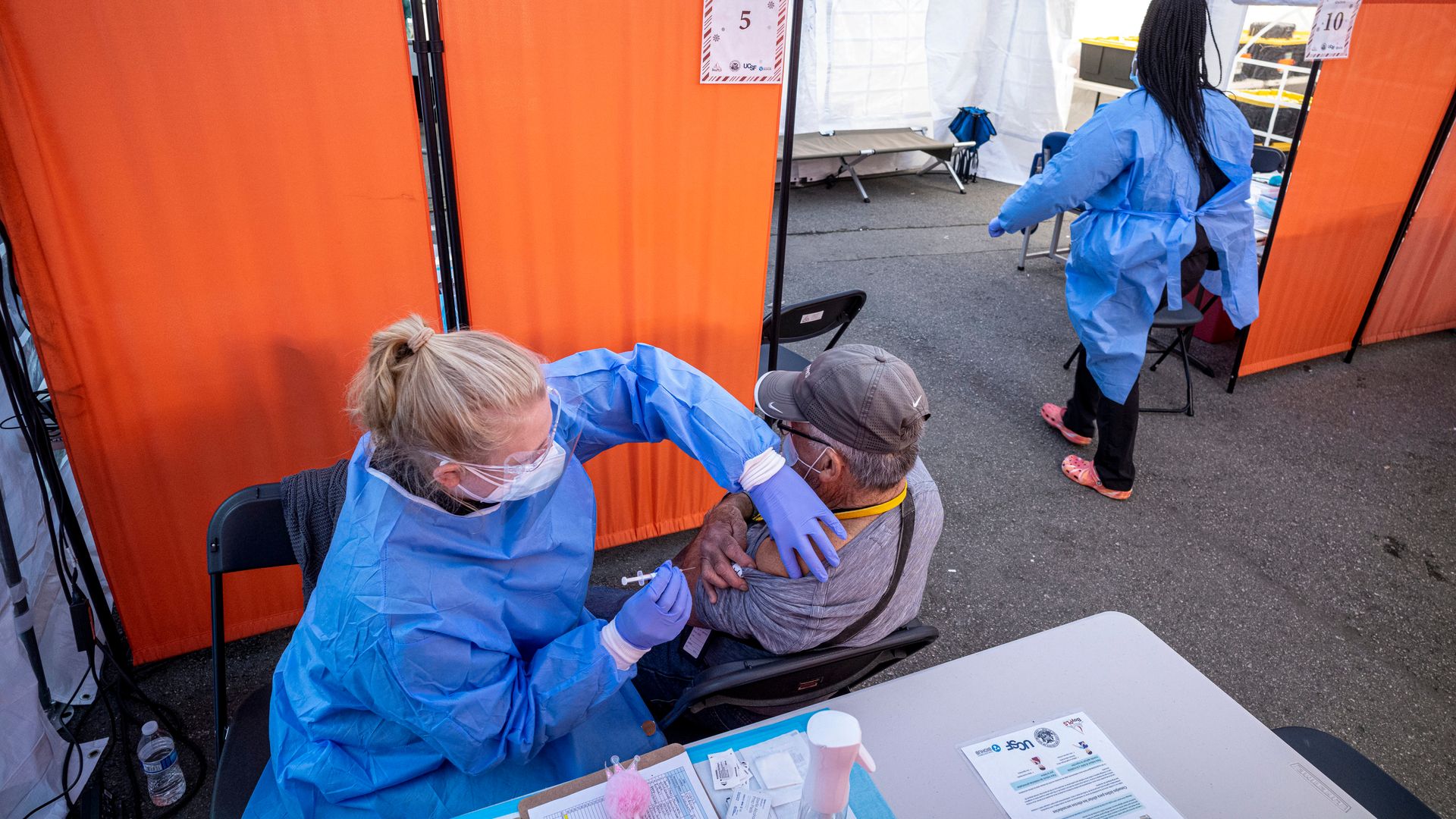
A health care worker administers a Pfizer COVID-19 vaccine in San Francisco on Jan. 10. Photo: David Paul Morris/Bloomberg via Getty Images
It's been over a year and a half since the first COVID-19 vaccines were administered in the U.S. In the months since, vaccine eligibility has changed, additional doses have been recommended and more shots have been delivered.
The big picture: An initial two-shot series is considered "fully vaccinated," but boosters are recommended for many age groups to be considered "up to date." Here's the latest on which type of COVID vaccine you're eligible to get and when.
Initial vaccine
- Adults 18 or older: People in this age group can receive a two-dose series of Pfizer or Moderna's COVID vaccine or a single dose of the shot developed by Johnson & Johnson.
- The CDC recommends that people in this age group who are moderately or severely immunocompromised receive a three-dose series of an mRNA vaccine — either from Pfizer or Moderna — and a one-shot booster after three months.
- People in this age group who are immunocompromised and initially received a Johnson & Johnson vaccine should get a second dose of either mRNA shot, the CDC said.
- All adults 18 and older who have received a primary series of their vaccine are recommended to get a booster.
Adolescents 12 to 17 years old: Adolescents in this age group may get an initial two-dose series of Pfizer's COVID vaccine.
- The CDC recommends that adolescents in this age group who are moderately or severely immunocompromised receive a three-dose series of Pfizer's vaccine and a one-shot booster after three months.
- Vaccines developed by Moderna and Johnson & Johnson are not authorized for this group.
Children 5 to 11 years old: Children in this age group are eligible to receive the Pfizer COVID vaccine but boosters are not available at this time, per the CDC.
- The CDC recommends that children in this age group who are moderately or severely immunocompromised receive a three-dose series of Pfizer's vaccine.
- Vaccines developed by Moderna and Johnson & Johnson are not authorized for this group.
Children under 5: Kids under 5 years old are not yet eligible to receive a COVID vaccine.
- Moderna has submitted a request to the FDA for emergency use authorization for its COVID vaccine in children six months to under six years of age.
- If approved, Moderna would be the first manufacturer to authorize a coronavirus vaccine for the youngest Americans.
First booster
Adults 18 and older: Everyone in this age group can get a booster of either Pfizer-BioNTech or Moderna at least five months after the final dose in the initial vaccine series.
- People who got the Johnson & Johnson vaccine should get a Pfizer or Moderna booster two months after the initial dose, the CDC said.
Adolescents 12 to 17 years old: Adolescents ages 12 to 17 are eligible to receive a booster of the Pfizer vaccine.
- Moderately or severely immunocompromised children and teens are eligible to receive the first booster at least three months after the final shot in their primary three-dose series, per the CDC.
Children 5 to 11 years old: The FDA has authorized booster shots of the Pfizer vaccine for this age group. The CDC still needs to sign off on the extra dose.
Second booster
Adults 50 years or older: Adults ages 50 or older are eligible to receive a second booster shot from either Pfizer or Moderna.
Adults 18 to 49: In this age group, people with compromised immune systems are currently eligible for an additional shot from either mRNA vaccine.
- People who received two doses of the Johnson & Johnson vaccine are also eligible for another shot of an mRNA vaccine.
Adolescents 12 to 17: People in this age group with compromised immune systems are eligible for a fourth shot. Only Pfizer's vaccine is authorized for adolescents between 12 to 17 years old.
Go deeper ... COVID-19 dashboard
Editor's note: This article has been updated with the FDA's approval of Pfizer's booster shot for children aged 5 to 11.
