Tuesday's health stories

FDA approves narrower use for drug floated for autism
The Food and Drug Administration on Tuesday approved using the drug leucovorin for a rare neurological condition — and not as a treatment for autism symptoms, as President Trump's health officials had suggested in September.
Why it matters: The label update dials back expectations that there could be a new autism remedy.
- It also represents a significant narrowing of the potential Trump administration officials had suggested for the little-known form of the vitamin folate.
Driving the news: The FDA has now approved the drug to treat cerebral folate deficiency, a condition that some people with autism also have but that is estimated to affect less than one in a million people.
Between the lines: That is well short of the potential promise for treating autism symptoms Trump administration officials suggested in September.
- FDA commissioner Marty Makary had suggested then that "hundreds of thousands" of children would benefit from the drug, and Health Secretary Robert F. Kennedy Jr. called it an "exciting therapy that may benefit large numbers of children who suffer from autism."
Yes, but: Though the FDA is not approving leucovorin for autism, "off-label" prescribing has surged following the White House event in September.
- A study published in The Lancet this month found a 71% increase in prescriptions after the White House briefing.
- Leucovorin is an existing drug that has traditionally been used alongside chemotherapy treatment.
- "This action may benefit some individuals with FOLR1- related cerebral folate transport deficiency who have developmental delays with autistic features," Makary said in a statement Tuesday.

FDA looks to boost biosimilars to lower drug costs
The Trump administration is moving to speed up approvals of copycat biologic drugs as a way to lower health costs and boost an underused market.
Why it matters: Efforts to elevate biosimilars have been thwarted by unfavorable placement on formularies and doctors' refusal to switch patients to the look-alike treatments.

Lawsuit seeks details on Medicare immigrant curbs
A senior citizen advocacy group is suing the Trump administration for details on how it's going to implement a new policy that prohibits certain legal immigrants from receiving Medicare benefits.
Why it matters: Last year's GOP tax-and-spending law stripped coverage from an estimated 100,000 immigrants covered by Medicare.

The states where hospitals are most concentrated
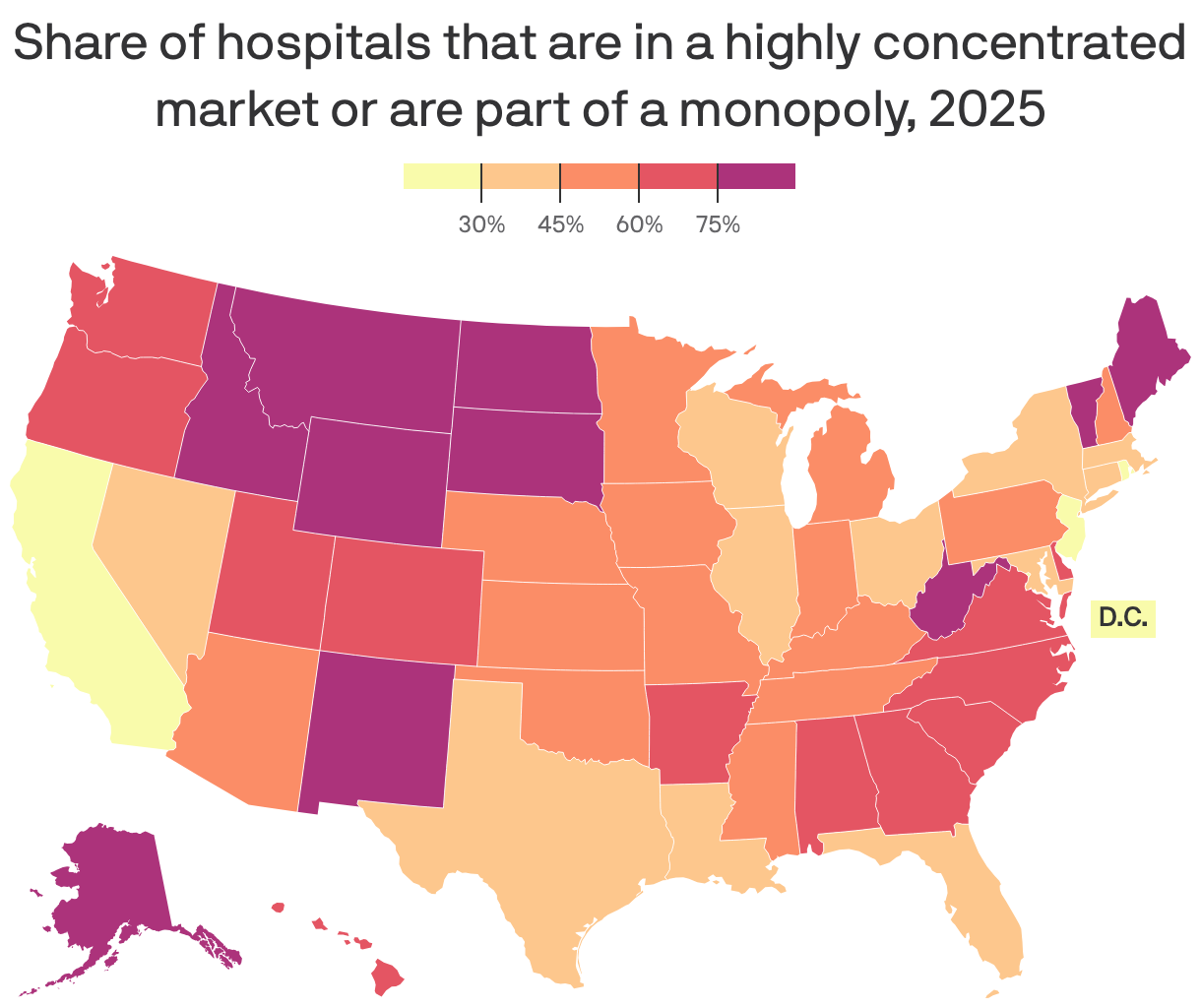
Every hospital in North Dakota, South Dakota and Wyoming is in a highly concentrated market, according to a blog post from Yale's new Health Care Affordability Lab.
Why it matters: As rising health care costs put more financial strain on Americans, "one major and underappreciated factor driving price increases is rising consolidation among U.S. hospitals," the blog post argues.

Congress plans new response to health cyberattacks
Two years after the seismic Change Healthcare cyberattack, Congress is advancing a plan to safeguard against the kind of hacks that can expose millions of people's private data and cripple health systems.
Why it matters: The bipartisan plan puts the burden on the government and providers to prevent the kind of breach that reverberates across the entire industry, jeopardizing patient access to needed treatments and costing hospitals billions.



