Scientists turn human skin cells into "functional" eggs
Add Axios as your preferred source to
see more of our stories on Google.
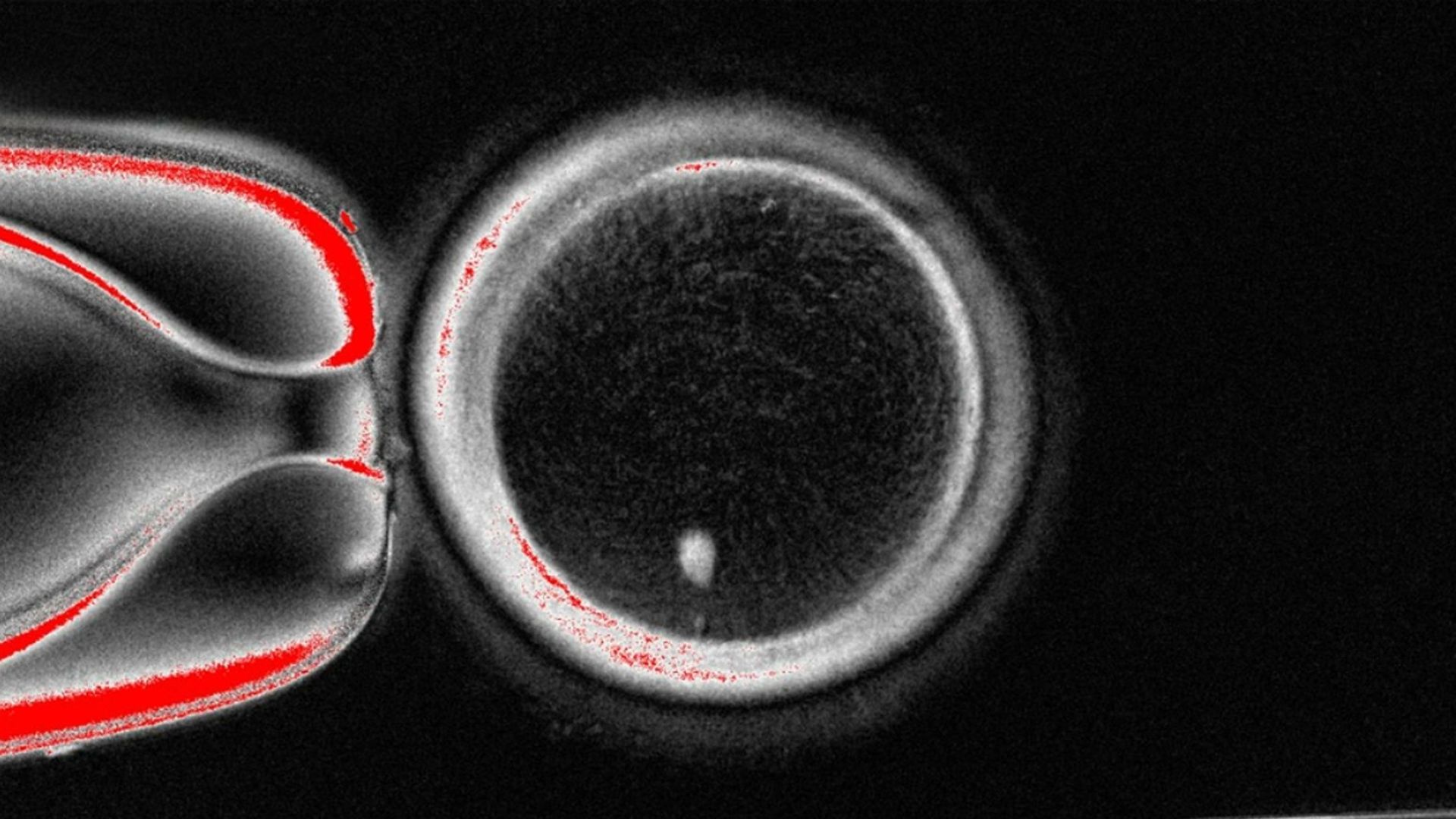
An image of an oocyte, or egg, with a bright image of a skin cell nucleus before fertilization. Image: Oregon Health & Science University
Scientists have turned DNA from human skin cells into "functional" eggs that are capable of producing early-stage embryos, according to a new study that cautions more research is needed as they're not yet usable.
Why it matters: "In addition to offering hope for millions of people with infertility due to lack of eggs or sperm, this method would allow for the possibility of same-sex couples to have a child genetically related to both partners," said study co-author Paula Amato, professor of obstetrics and gynecology at Oregon Health & Science University, in a statement Tuesday.
- This early-stage research could one day also be used to treat infertility for women of advanced maternal age "or those who are unable to produce viable eggs due to previous treatment of cancer or other causes," according to an OHSU post.
Yes, but: The Portland-based team noted several limitations in their proof-of-concept study, published in Nature Communications on Tuesday, notably that all of the embryos had chromosomal abnormalities.
- And only 9% of the 82 eggs created in the Oregon lab developed six days after fertilization, when embryos are typically transferred to establish a pregnancy through IVF. None were cultured beyond that point.
- Still, senior study author Shoukhrat Mitalipov, director of the OHSU Center for Embryonic Cell and Gene Therapy, noted researchers have "achieved something that was thought to be impossible."
What they did: The early-stage research used a technique that involves moving a skin cell nucleus into a donor egg stripped of its nucleus, a processed pioneered in the cloning of Dolly the sheep in 1997.
- While Dolly was created after the cloning of one parent, the researchers' work resulted in embryos with chromosomes contributed from both parents.
Zoom in: Per the OHSU, the scientists undertook three steps to achieve the results:
1. Researchers moved the nucleus of a skin cell into an egg, or oocyte, stripped of its own nucleus.
2. Prompted by cytoplasm, a gelatinous liquid that fills the inside of a cell, within the donor egg, "the implanted skin cell nucleus ideally discards half of its chromosomes in a process similar to meiosis (cell division)."
- This is a crucial step that results in a haploid egg, the presence of a single set of chromosomes in an organism's cells, with a single set of 23 chromosomes rather than 46 that humans have, inheriting 23 pairs from each parent.
3. Researchers fertilized the new egg with sperm through the standard IVF process, creating a diploid embryo, or the presence of two complete sets of chromosomes.
Between the lines: "For the first time, scientists have shown that DNA from ordinary body cells can be placed into an egg, activated, and made to halve its chromosomes, mimicking the special steps that normally create eggs and sperm," said Ying Cheong, a professor of reproductive medicine at the University of Southampton, in a statement.
- "This breakthrough, called mitomeiosis, is an exciting proof of concept," said Cheong, of the pioneering technique researchers used to mimic natural cell division.
- "While this is still very early laboratory work, in the future it could transform how we understand infertility and miscarriage, and perhaps one day open the door to creating egg- or sperm-like cells for those who have no other options," added Cheong, who was not involved in the study.
- Richard Anderson, a professor of clinical reproductive science at the University of Edinburgh, said in a statement the "ability to generate new eggs would be a major advance, and this study shows that the genetic material from skin cells can be used to generate an egg-like cell with the right number of chromosomes to be fertilised and develop into an early embryo."
- Anderson, who wasn't involved in the study, said while there "will be very important safety concerns," it "is a step towards helping many women have their own genetic children."
What's next: Researchers "expect at least a decade of further research before the approach could be deemed safe or effective enough to advance to a clinical trial, even assuming such a trial would be permitted in the United States," per a statement posted on the OHSU's website.
- The study notes that while the research shows potential, "further research is required to ensure efficacy and safety before future clinical applications."
Go deeper: The future of fertility
