Supreme Court preserves abortion pill access after tossing challenge
Add Axios as your preferred source to
see more of our stories on Google.
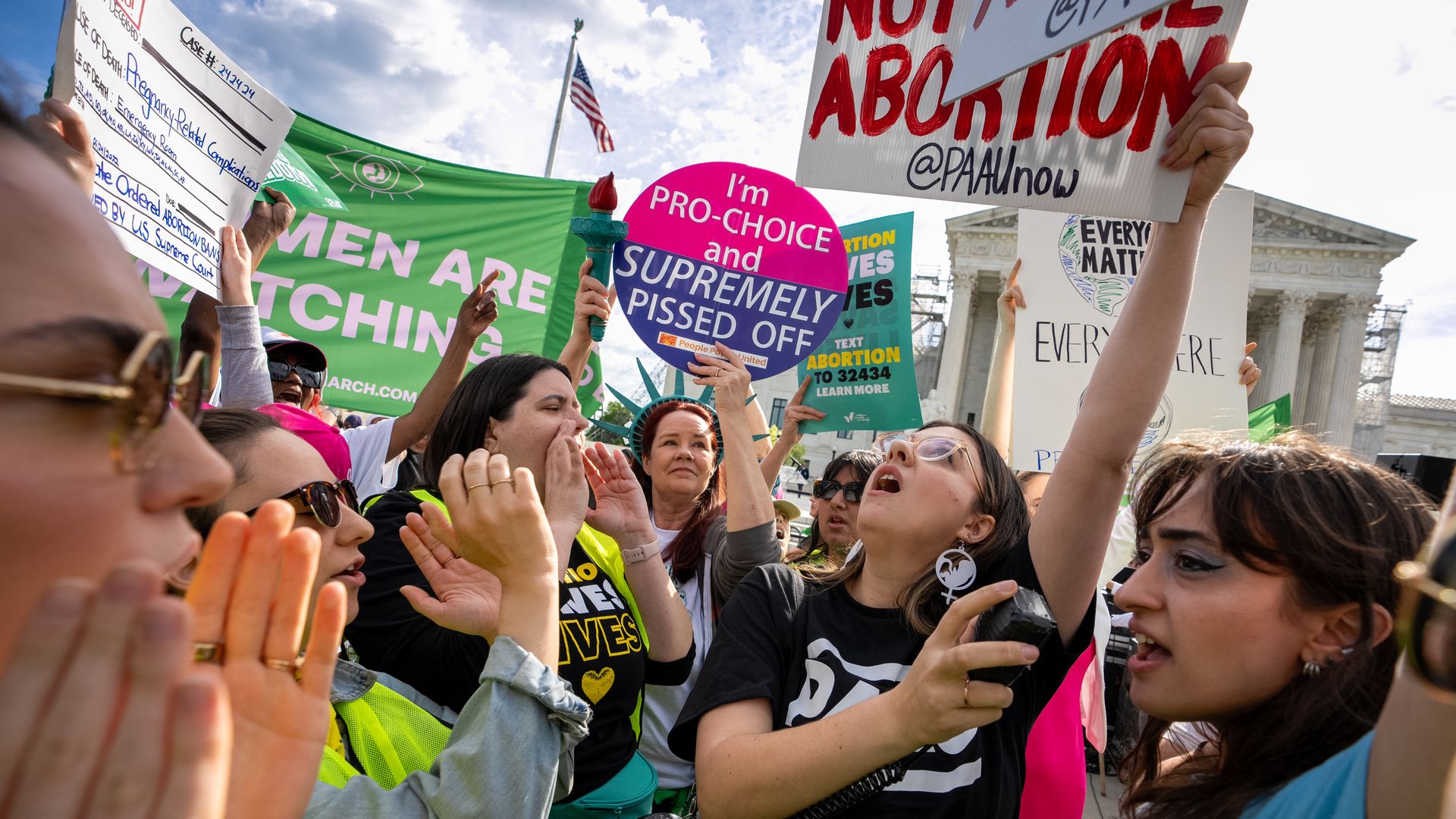
Abortion-rights and anti-abortion supporters clash outside the Supreme Court on April 24. Photo: Andrew Harnik/Getty Images
The U.S. Supreme Court threw out its biggest abortion-related case since overturning Roe v. Wade two years ago, saying Thursday that doctors opposed to a commonly used abortion pill lacked legal standing.
Why it matters: Rejecting the highly anticipated case preserves access to mifepristone but allowed the court to skirt making a ruling on the merits of the case.
- The Supreme Court held that the plaintiffs — a group of doctors who oppose abortion but don't take or prescribe mifepristone — couldn't show they were directly injured, as the Biden administration asserted during oral arguments.
Zoom in: The plaintiffs' argument would lead courts down an "uncharted path" that would allow citizens to challenge "virtually every government action that they do not like," Justice Brett Kavanaugh wrote in the unanimous decision.
- "Doctors have never had standing to challenge FDA's drug approvals simply on the theory that use of the drugs by others may cause more visits to doctors," he wrote.
- The court compared this to other fields: "Firefighters could sue to object to relaxed building codes that increase fire risks. Police officers could sue to challenge a government decision to legalize certain activities that are associated with increased crime."
The other side: "While we're disappointed with the court's decision, we will continue to advocate for women and work to restore commonsense safeguards for abortion drugs," Alliance Defending Freedom senior counsel Erin Hawley said in a statement.
The big picture: Medication abortion accounts for 63% of abortions in the U.S., per the Guttmacher Institute, a research organization that supports abortion rights.
- Mifepristone is commonly used in tandem with a second FDA-approved drug, misoprostol, in the first 10 weeks of pregnancy. Both pills have long safety and efficacy records.
- The pills are increasingly prescribed online and mailed to patients after the FDA eliminated the requirement that they be picked up in person.
- Walgreens and CVS announced plans in March to start selling abortion pills in states where the procedure is legal.
Context: The federal government argued during oral arguments in March that the group of doctors who objected to abortion and brought the case against the FDA didn't have the legal standing to do so.
- Solicitor General Elizabeth Prelogar argued that the doctors don't prescribe mifepristone and haven't suffered any real injury from the FDA's regulatory decisions.
- The doctors argued that the injury is the time spent in the emergency room treating women who have taken the drug.
What they're saying: The Supreme Court's ruling maintains the stability of the FDA's drug approval process, Abigail Long, a spokesperson for abortion pill distributor Danco Laboratories, said in a statement.
- "Danco is proud to have led the way in obtaining FDA approval of this essential reproductive health option and of modifications to the conditions under which it can be used," Long said.
- "We thank the Court for its careful analysis and remain committed to developing and bringing to market safe and effective products in this crucial area of public health."
Catch up quick: The 5th U.S. Circuit Court of Appeals found the FDA failed to take into account safety concerns last August when it expanded the time frame in which mifepristone could be used in pregnancy and when it dropped a requirement that the pills be prescribed in person.
- The three-judge panel on the appeals court partially blocked a lower court ruling that struck the FDA's original 2000 approval of the drug in response to a suit from anti-abortion forces.
- The maker of mifepristone, Danco Laboratories, and the Department of Justice asked justices to review the 5th Circuit ruling, and the Supreme Court agreed in December to review the decision.
Go deeper:
- SCOTUS abortion pill hearing puts FDA discretion on trial
- Abortion provider protections prove crucial in a post-Roe world
- Where abortion is on the ballot in November
Editor's note: This story has been updated with details from the decision and comments from Danco Laboratories and the Alliance Defending Freedom senior counsel Erin Hawley.

