Jul 17, 2023 - Health
Medicare proposes broader coverage of PET scans for Alzheimer's patients
Add Axios as your preferred source to
see more of our stories on Google.
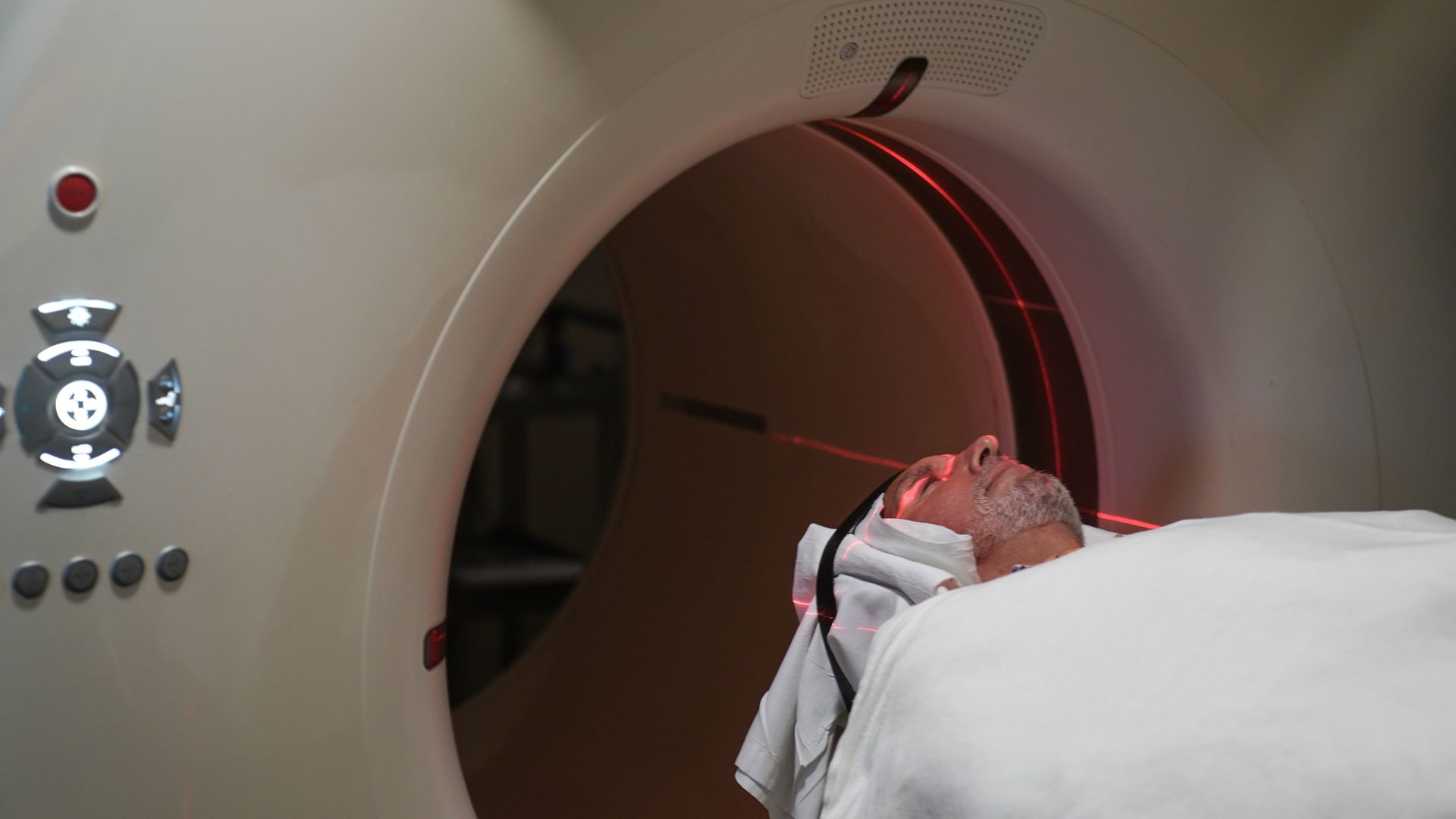
Jay Reinstein, who suffers from Alzheimer's, prepares to receive a PET scan at MedStar Georgetown University Hospital in Washington, DC on June 20, 2023. Photo: Michael Robinson Chávez/The Washington Post via Getty Images

