Study finds sharp drop in FDA inspections since pandemic began
Add Axios as your preferred source to
see more of our stories on Google.
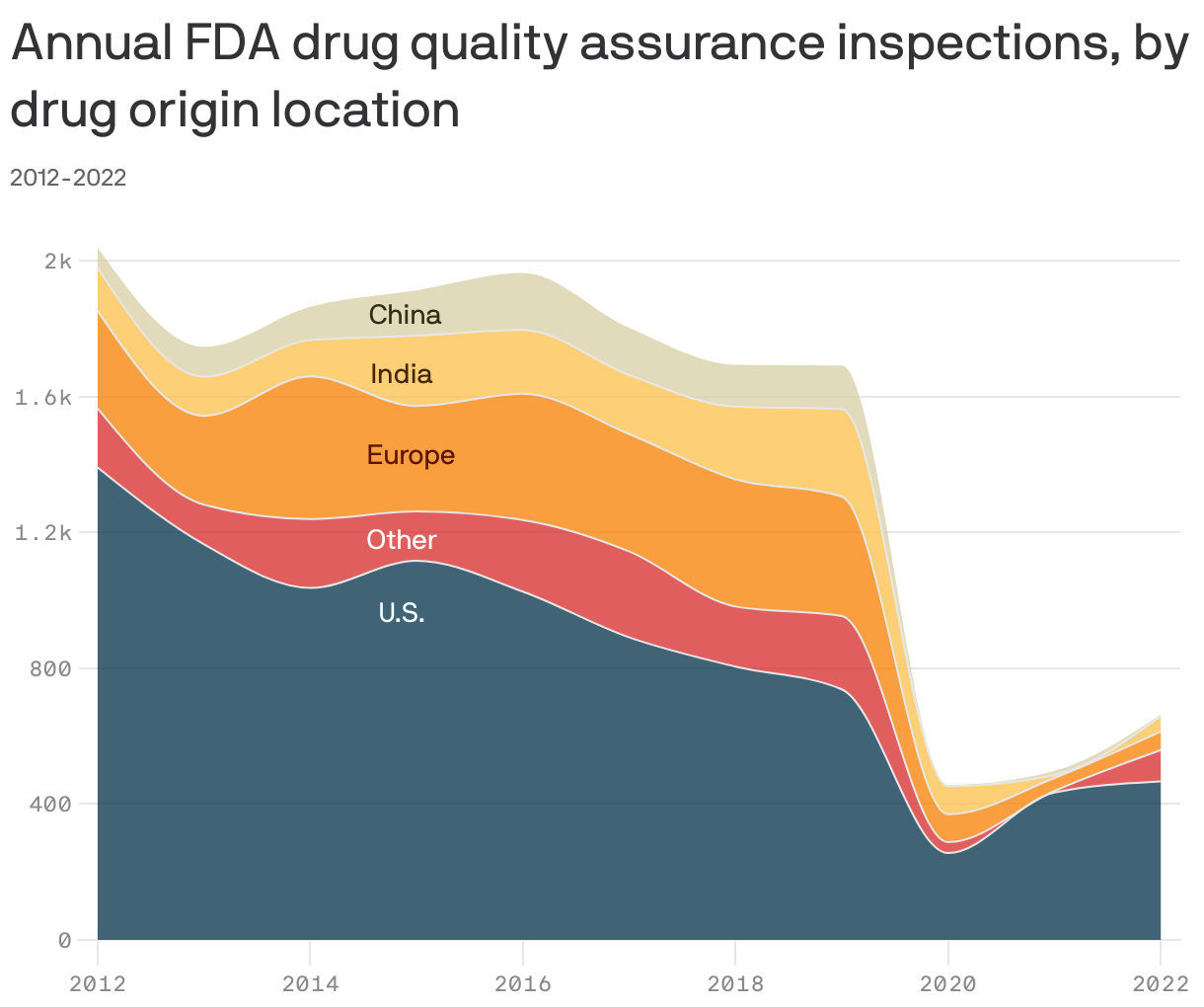
Food and Drug Administration inspections of drug manufacturing facilities in the U.S. and abroad dropped well below pre-pandemic levels between 2020 and 2022, according to a new study in Health Affairs.
Why it matters: The findings are further evidence of a fragile global drug supply chain at a time when some critical medicines are in short supply. Some of the squeeze was due to inspections that took overseas facilities offline because of safety concerns.
Between the lines: The FDA early in the pandemic said it would curtail many of its inspections, but work picked back up by July 2020. However, the agency didn't resume nonprioritized domestic inspections and non-mission-critical foreign inspections until last year, the authors write.
- "As the world moves beyond the pandemic, it is crucial for the FDA to address the backlog of inspections and to consider new approaches for the future," they wrote.
By the numbers: The study found a 79% decrease in inspections of foreign facilities and a 35% drop in inspections of domestic facilities in 2022 compared to 2019.
- "Notably, the FDA conducted no inspections in China or India between April 2020 and March 2021," the authors wrote.
- The study found no corresponding reduction in drug manufacturing or imports. The researchers also found citations rose dramatically.
What they're saying: "The American public should rest assured that the drugs they rely on are high quality, safe and effective," the FDA said in a statement. "Protecting patients is the highest priority of the FDA and the agency remains committed to using all available tools to oversee the safety, effectiveness and quality of FDA-regulated drug products."
Editor's note: This story was updated with FDA comment.
