Axios Vitals

January 04, 2023
Hey, it's already Wednesday, Vitals readers! Today's newsletter is 917 words or a 3½-minute read.
📲 1 quick thing: All that time your kids are spending on social media could be affecting how they respond to social rewards and punishments, according to a study published Tuesday in JAMA Pediatrics.
- A study of 169 sixth and seventh-graders suggests social media checking behaviors in early adolescence is associated with changes in the brain's sensitivity to peer feedback, per the New York Times. (Put down the phones, kids...)
1 big thing: FDA adds a major twist to the abortion pill fight
Illustration: Maura Losch/Axios
The FDA yesterday added a new twist to the escalating fight over medication abortion by allowing pharmacies to offer the pills — even as anti-abortion groups mount a unique legal challenge to the approved use of the drugs, Axios' Oriana Gonzalez writes.
The big picture: The developments underscore how much access to mifepristone, which is used in first-trimester abortions, has become a focal point in the reproductive health wars since the Supreme Court struck down precedents that established a federal right to abortion.
- Medication abortion accounts for over half of abortions in the country.
- A Trump-appointed judge as early as next month will decide whether to grant a request to temporarily freeze the FDA's 2000 approval of mifepristone, which could end nationwide access to the drug.
Driving the news: The FDA yesterday made a regulatory change that may allow retail pharmacies to offer abortion pills, effectively expanding access as more red states move to ban or restrict access to the procedure.
- With the change, the FDA has officially removed mifepristone's in-person dispensing requirement and allowed for pharmacies to seek certification to offer the medication.
- However, pharmacies might still need to comply with the laws of the state they are located in, some of which could restrict the ability to provide such pills.
What they are saying: A CVS spokesperson said Tuesday night that the chain is evaluating requirements to dispense the drug in states that don't restrict the availability of abortion pills. A Walgreens spokesperson said the company is reviewing the policy change.
Between the lines: The conservative group Alliance Defending Freedom filed a lawsuit in November on behalf of the Alliance for Hippocratic Medicine — which is comprised of several anti-abortion groups — arguing that the FDA did not properly approve mifepristone for terminating pregnancies.
- "This case and this litigation is now ripe to go to court, where we can now challenge everything that the FDA has done," said Erik Baptist, the Alliance Defending Freedom senior counsel.
What we're watching: Based on the court schedule, a decision on whether to grant the request to temporarily block the FDA approval could come as early as February.
2. Medicare Part B oversight gaps costs millions
Medicare and its enrollees were unable to realize millions in savings because of gaps in CMS' oversight of the Part B program, according to a federal watchdog report released Tuesday.
Driving the news: The federal health department's Office of the Inspector General looked at oversight of manufacturer-reported data on average sales prices (ASP). When that data is inaccurate, Medicare may make inappropriate payments, the report said.
By the numbers: OIG found Medicare administrators could not calculate an ASP-based payment amount for 8% of drug codes at least once between 2016 and 2020.
- Nearly a quarter of drug codes were missing ASP data for one or more specific drugs within that code in at least one quarter during that span.
What they're saying: "Given the cost and importance of these life-saving drugs, it is vital that CMS conduct robust oversight to ensure that Medicare makes appropriate payments for Part B-covered drugs," OIG wrote.
- CMS said it "believes ASP reporting is consistent with statutory requirements and results in the determination of accurate payment limits, but that it shares OIG's concerns," the report said.
3. Health deals cooled off last year
Illustration: Aïda Amer/Axios
Health care M&A deal value dropped off significantly last year to $328.8 million, down nearly 35% from $504.9 million in 2021, Axios' Claire Rychlewski writes from newly released Refinitiv data.
- Yet the sector's share of the overall dealmaking market inched higher.
Why it matters: Recession fears spotlight sectors' vulnerability to economic downturns. But investors argue, correctly, that everybody gets sick, Claire writes.
- Last year there were 4,523 health care deals, down about 25% from 6,070 deals in 2021.
In what was otherwise a slow year, companies like health care distributors were the big winners, the Wall Street Journal wrote. "In a year of stock market turmoil, healthcare investors flocked to boring but steady," wrote
Between the lines: Pharma giants GSK, Amgen and J&J made the list of top deals worldwide.
- Health care providers and services accounted for $60.6 million of dealmaking activity last year, down 11.6% from about $68.6 million in 2021.
What to watch: Health care deals are "set to surge" in 2023, at least in the pharma sector, with activity picking up in the last couple of months, the WSJ wrote, saying likely deal makers include J&J, Pfizer and Merck.
- Market conditions are also good for increased digital health consolidation, Fierce Healthcare reported.
⚡️ 4. Lightning round: New U.S. health laws
The new year has brought a wide range of new state health laws.
- New York passed a law to cover extra costs associated with organ donations. The Empire State has also started requiring every private insurance plan offering maternity care coverage to cover abortion.
- California now has a law guaranteeing a person's "fundamental right" to abortion while Tennessee has a new law that requires "abortion-inducing drugs" be provided by physicians in medical facilities, not through the mail or pharmacies.
- Maryland is capping the price of insulin for anyone on state-regulated insurance plans.
- Colorado will decriminalize psilocybin-containing mushrooms and other psychedelic drugs starting today for people over 21. Oregon became the first state to allow adult use of mushrooms on Sunday.
5. Catch up quick
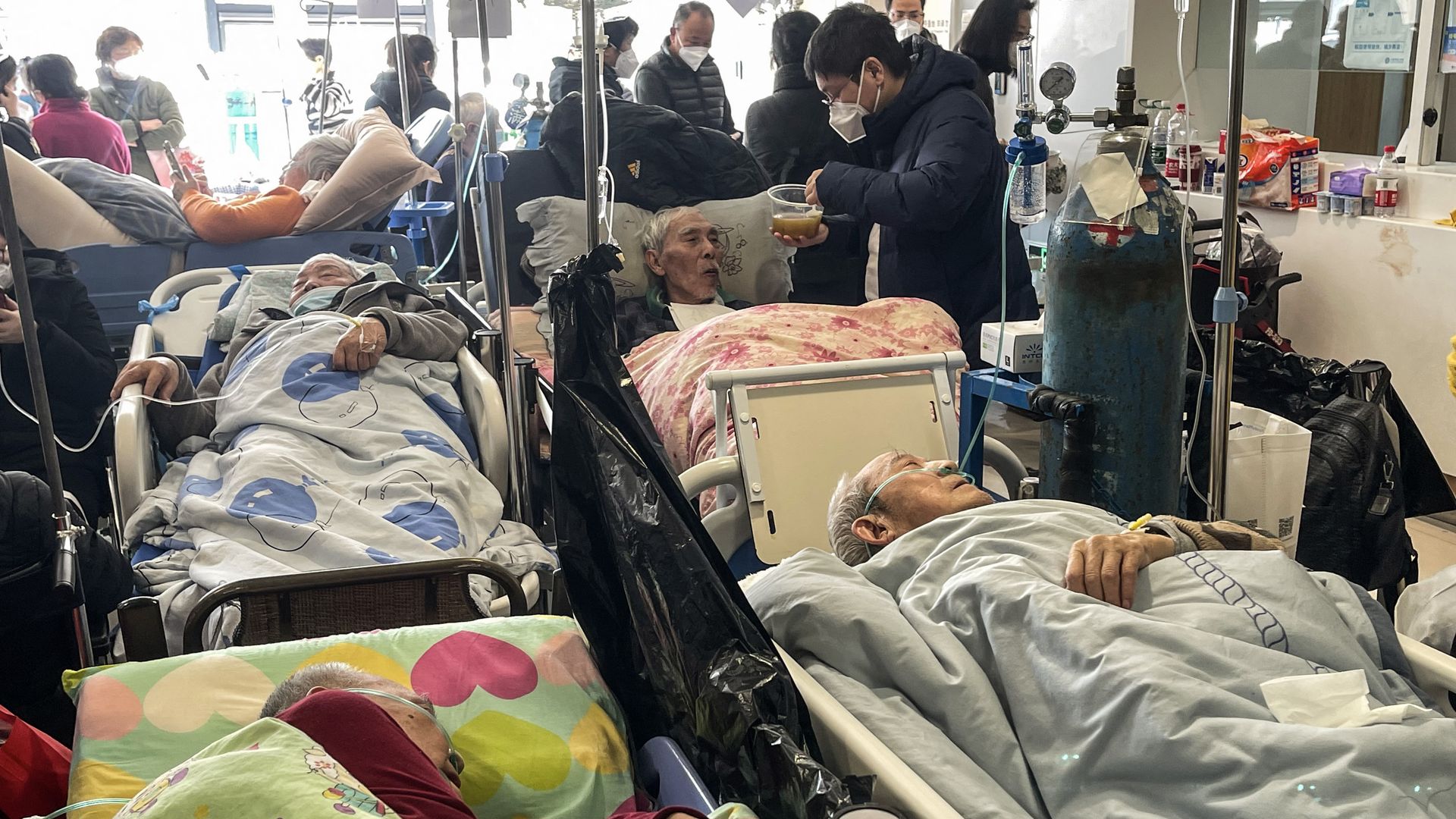
Patients on stretchers are seen at Tongren hospital in Shanghai on Jan 3. Photo: Hector Retamal/AFP via Getty Images
🏥 An enormous wave of COVID infections is straining China's medical facilities, as Chinese state media said authorities had implemented "a planned, science-based approach." (Axios)
🦠 What we know and don't know about the XBB.1.5 COVID subvariant. (Axios)
👉 How antibody drugs could target infectious diseases — if costs come down. (Washington Post)
Did someone forward this email to you? Here's how to sign up.
Thanks for reading, and thanks to senior editor Adriel Bettelheim and senior copy editor Bryan McBournie for the edits.
Sign up for Axios Vitals

Healthcare policy and business analysis from Tina Reed, Maya Goldman, and Caitlin Owens.