Breaking through the brain's barrier
Add Axios as your preferred source to
see more of our stories on Google.
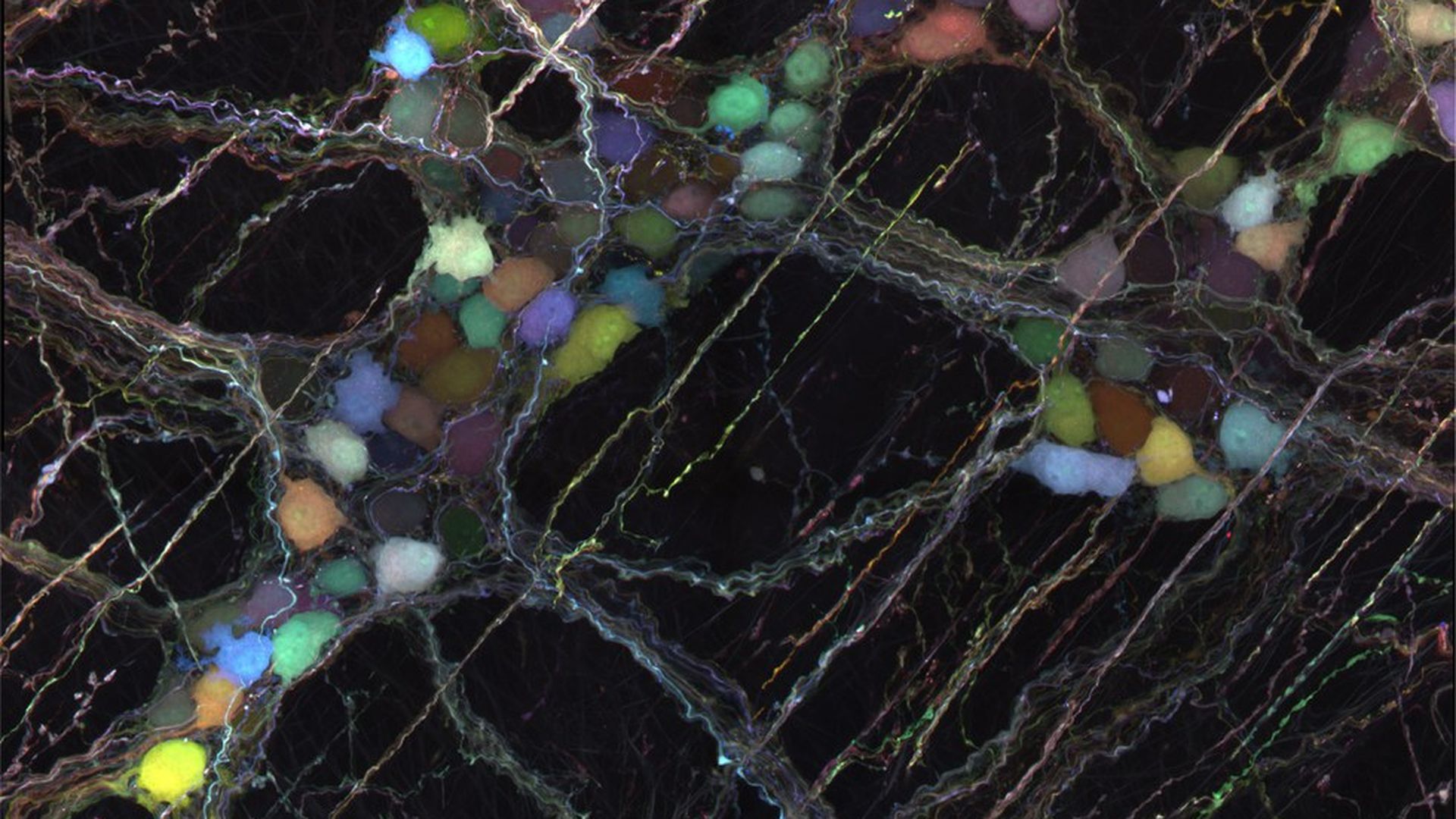
Caltech
A strong defense system allows what the brain needs in and keeps out toxins and pathogens. It's a vital function that also stops promising drugs from reaching their target (think: a tumor) and prevents neuroscientists from probing how the brain works. Scientists at Caltech have now engineered viruses that can bypass this barrier in mice and deliver genes to many different types of cells.
Why it matters: In the past few decades, a wealth of tools have been created to study the brain — neurons can be individually labeled and imaged, genes can conceivably be edited, and the brain's cells can be manipulated with light in order to see what effect it has on functions like memory. But that all hinges on getting into the brain. This new work opens up the brain's intricacies for unprecedented study and possibly treatment of neural disorders (like Parkinson's and Huntington's diseases), brain cancers, and Friedreich's ataxia and other peripheral nervous system diseases.
The incidence of brain diseases is on the rise, mainly because of an aging population, but drugs have come up short because they can't cross the blood brain barrier in adequate amounts to be effective. Investigators are trying to shepherd drugs in on molecules that mimic venom, deliver them with ultrasound, or use quantum dots to make it past the barrier.
"We increasingly know what is going wrong in brain disorders, and have new genetic tools that could correct the problems, but delivery of the tools to the right parts of the brain remains a challenge," says USC's Andrew Hires, who wasn't involved in the study.
What they did: The main function of a virus is to deliver its genes into cells. Scientists have successfully leveraged that by reprogramming viruses to deliver genes into cells of choice — but the challenge is that it takes a lot of viruses to deliver that cargo to hard-to-reach regions of the brain and throughout the nervous system.
Last year, Viviana Gradinaru and Ben Deverman engineered a naturally occurring virus that can move through the blood-brain barrier in mice, and in a paper published last month reported they engineered the shell of that virus to create two variations that can broadly deliver genes to the nervous system:
- One can get its cargo into striatal neurons deep within the brain with 55% efficiency and to cortical neurons with 69% efficiency using a relatively low dose of the virus. (Previous work suggests a higher virus dose would produce higher efficiencies, the researchers say.)
- The second engineered virus can target neurons outside of the brain and spinal cord. There is a vast peripheral nervous system touching the heart, gut, lungs, pancreas and other organs that senses pain and regulates breathing, digestion and heart rate.
How it works: The virus is injected into the bloodstream, carried into the brain and through the peripheral nervous system, where it is picked up by neurons. Once inside, the virus releases its genetic cargo, which the cell then begins to decipher and express. That cargo could be a functioning copy of a gene that could replace a mutated one in someone with a disease like Huntington's or a fluorescent protein that can be used to track cells.
"[The viruses] are plug and play with whatever is trying to be delivered," says Deverman. Whether for testing therapies, studying how a disease develops, gene-editing or manipulating neurons with optogenetics, "they provides efficient access to the peripheral nervous system and the central nervous system."
- This is an advance in that past studies called for the creation of genetically modified animals (a process of months to years) or invasive surgeries. Delivering therapies to the brain requires opening the skull and the brain's protective sheath.
"These viruses should be very useful for research on the basic functions of the brain, and for translating these findings into new therapies. For example, these viruses could help us understand the logic of pain processing circuitry in the spinal cord and brain. Then they could potentially allow precise adjustment of the activity patterns within these circuits to relieve chronic pain without the use of highly addictive opioid drugs," says Hires.
What's next: The work so far has been done in mice — whether the blood-brain barrier in the animals differs from that in humans in relevant ways will require further study. The adeno-associated viruses the researchers use are known to humans, meaning our immune systems could recognize and destroy them. The researchers are looking at whether that too can be solved with some engineering. Hires cautions: "Showing safety and efficacy in primates is an important next step before we see this technology in the clinic."
