Clusters of lab-grown cells show role of key genes in brain disorders
Add Axios as your preferred source to
see more of our stories on Google.
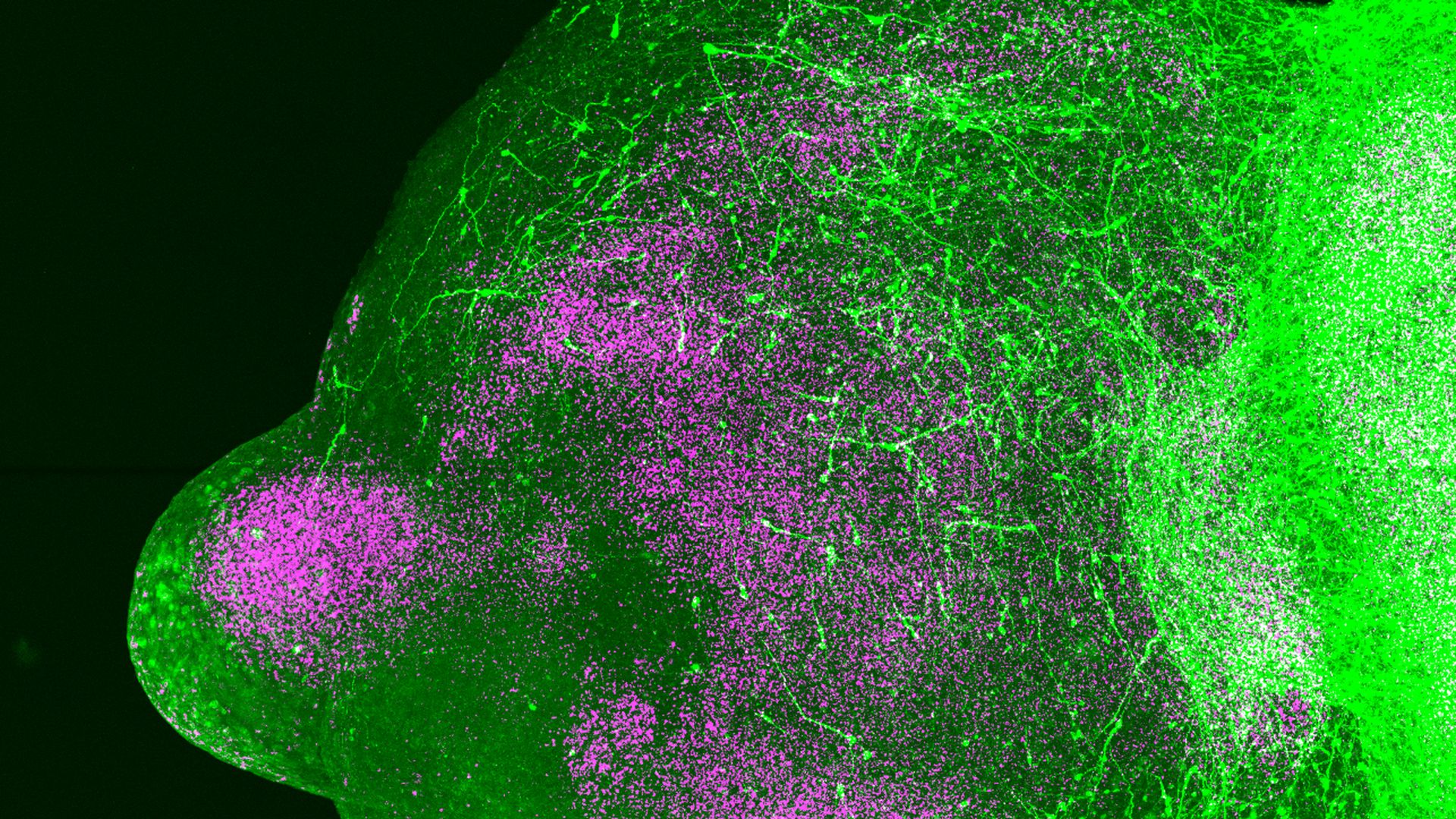
Neurons migrating in an assembloid. Image: Pasca Lab, Stanford University
A combination of gene editing and growing tiny clusters of brain cells in the lab can be used to figure out how certain genes are involved in the brain's development and disorders, researchers reported.
Why it matters: Mutations in hundreds of genes are known to be associated with neurodevelopmental disorders, but how they affect the function of neurons is largely unknown. Understanding their impact could inform therapies that target the genes and the processes they affect.
How it works: During the brain's development, cells called interneurons take on different roles and migrate through the brain to their correct location, forming circuits that regulate brain processes.
- Scientists have hypothesized that the balance in signaling between interneurons that excite or inhibit the firing of other neurons in the circuit is perturbed when people develop autism, schizophrenia and epilepsy.
- The imbalance may also underlie schizophrenia, epilepsy and other disorders.
What they did: Sergiu Pasca, a neurobiologist at Stanford University, and his colleagues leveraged CRISPR gene editing and the ability to grow brain organoids to study the role of interneurons.
- Using techniques from earlier work, the team grew stem cells into brain organoids representing the subpallium (a region deep in the brain where the majority of the brain's inhibitory cells originate) and the cortex.
- When they are put together, the cells start moving toward the cortex and start populating it exactly as they would do in utero, Pasca says.
- In the new study, published this week in Nature, the team made more than 1,000 subpallium and cortex organoids and used CRISPR gene editing to knock out the function of one of 425 different genes associated with autism in each organoid.
- They then put the organoids together and, using a fluorescent dye, watched how the neurons moved in the dish.
What they found: About 10% of the 400-plus genes interfere with the generation of the interneurons or their migration during the brain's development, the researchers reported.
- "A few were complete surprises," Pasca says. A gene associated with a severe but rare disease affecting children affected an organelle in cells and disrupted the cells' migration in the brain.
Yes, but: There are other processes that genes could interfere with at later stages of development and weren't looked at in the study.
- Another challenge is "rare neurodevelopmental disorder genes don't act in isolation," says Kristen Brennand, a professor of psychiatry and of genetics at Yale University who wasn't involved in the study.
- "Three different people can inherit the same risk gene, and one will have autism, another schizophrenia, and a third be unaffected. Repeating the screen across more donors will help explain these variable effects."
The big picture: The next step is to generate similar screens for other processes in the brain that could be disrupted by disorders and disease, including how cells mature, how cells take on their different types, how they establish synapses, Pasca says.
- Understanding these processes could offer a new way of looking at psychiatric disorders that are currently defined through the lens of behavior, not biology, he says.
Of note: Another study published this week used brain organoids and CRISPR and found the effects of 36 high-risk autism spectrum disorder genes on determining the fate of cells.t
