FDA authorizes updated COVID shots for kids as young as 6 months old
Add Axios as your preferred source to
see more of our stories on Google.
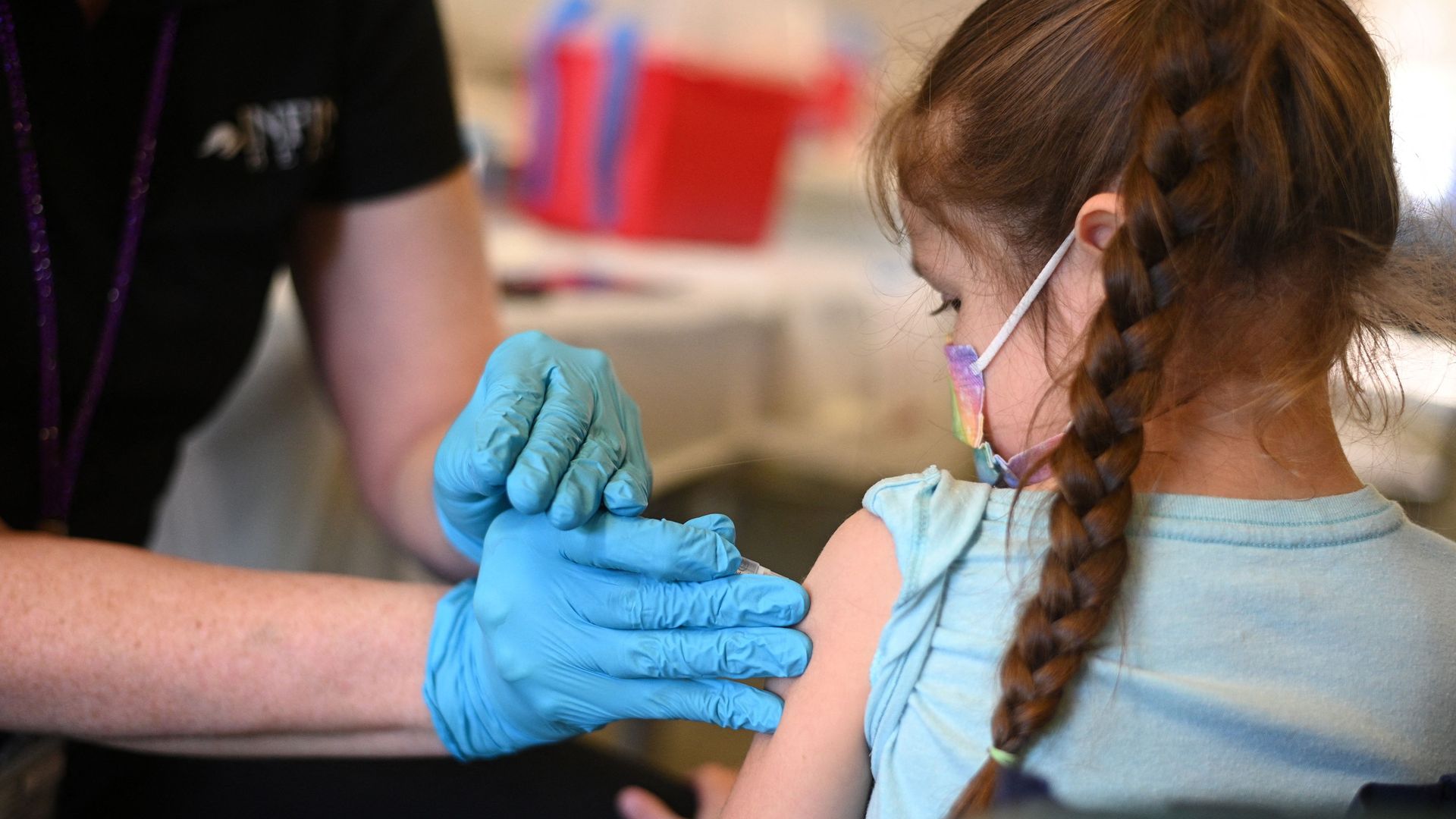
Photo: ROBYN BECK/AFP via Getty Images
The Food and Drug Administration on Thursday authorized updated COVID-19 shots from Pfizer and Moderna that target the omicron variant for children as young as six months.
Driving the news: FDA Commissioner Robert Califf said in a statement that it was important to get young children vaccinated ahead of the holidays, "where more time will be spent indoors."
The big picture: Health experts worry that new COVID variants could drive a surge of cases in the winter.
- The number of hospitalizations has increased following the Thanksgiving holiday, according to data from the Centers for Disease Control and Prevention.
State of play: Kids between six months and five years old who received the Moderna primary series can receive the updated Moderna booster as soon as two months later.
- Children between six months and four years old who received two doses of the Pfizer-BioNTech vaccine can get the companies' updated vaccine as a third shot.
- But those who received three doses of Pfizer's monovalent vaccine aren't eligible for an updated shot. The FDA said it is still evaluating data for young children in this group, and an update could come after January.
What they're saying: "Parents and caregivers can be assured that the FDA has taken a great deal of care in our review, and we encourage parents of children of any age who are eligible for primary vaccination or a bivalent COVID-19 vaccine booster dose to consider seeking vaccination now as it can potentially help protect them from COVID-19 during a time when cases are increasing," said Peter Marks, director of the FDA's Center for Biologics Evaluation and Research.
- Recent research showed bivalent mRNA vaccines provided additional protection against infection compared with previous vaccination with monovalent vaccines alone.
What's next: The CDC will evaluate the shots and must give approval before physicians can start administering them.
Go deeper: Biden faces COVID dilemma as a winter wave nears
Editor's note: This story was updated with context about COVID hospitalization data.
