Sep 26, 2022 - Health
Pfizer and BioNTech ask FDA to authorize updated COVID booster for children
Add Axios as your preferred source to
see more of our stories on Google.
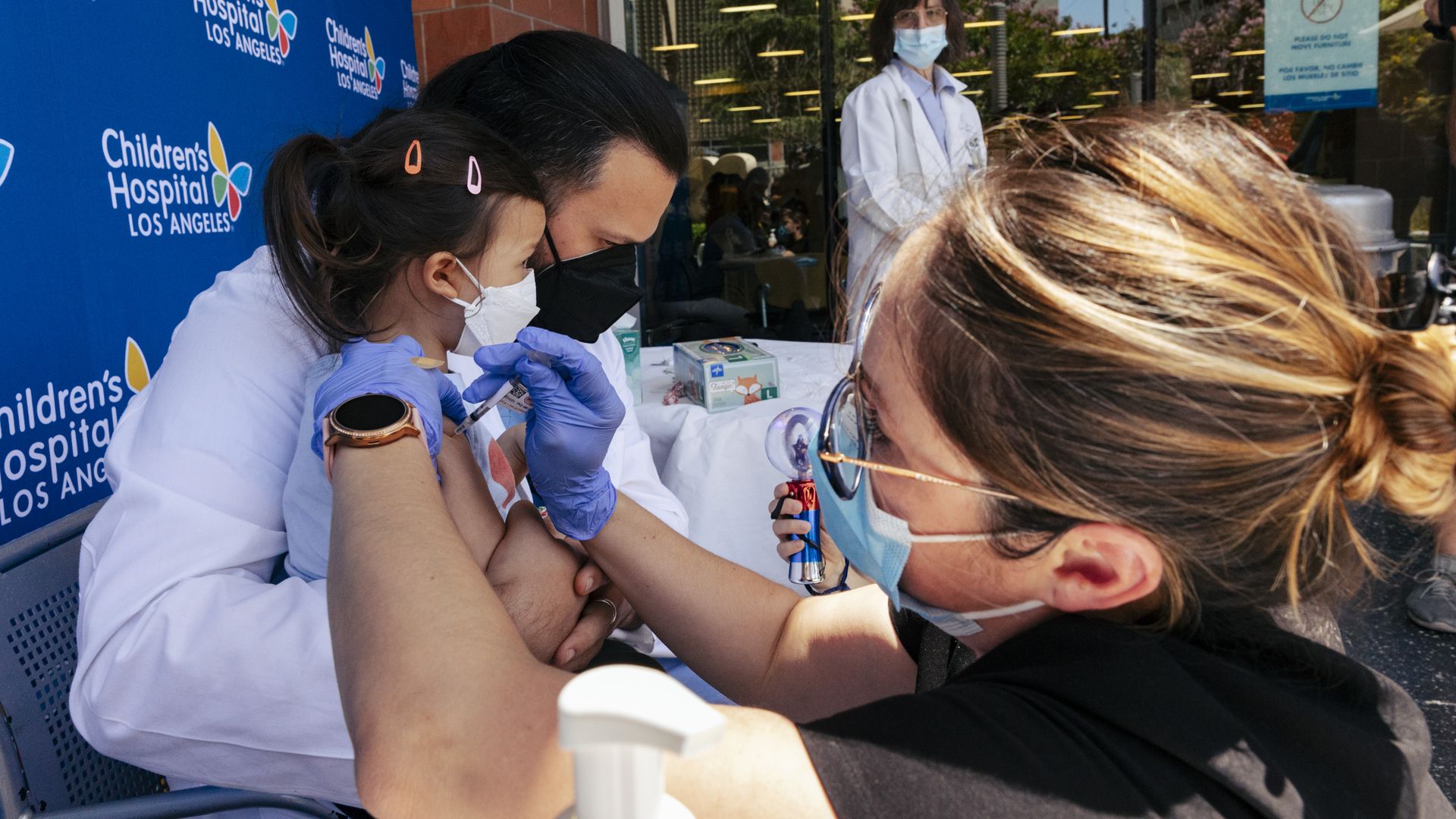
Sofia Espinoza Tam is held by her father, Dr. Juan Espinoza, while a nurse administers the Pfizer COVID-19 vaccine outside on June 21 in Los Angeles, CA. Photo: Wesley Lapointe / Los Angeles Times via Getty Images

