Monkeypox treatment enters Phase 3 clinical trial
Add Axios as your preferred source to
see more of our stories on Google.
A smallpox treatment that's become a backstop for patients who test positive for monkeypox in the U.S. is going into a Phase 3 clinical trial to test its effectiveness.
Why it matters: While preliminary CDC data show tecovirimat, called TPOXX, appears to be working to alleviate symptoms, only a late-stage trial will truly show how effective it is.
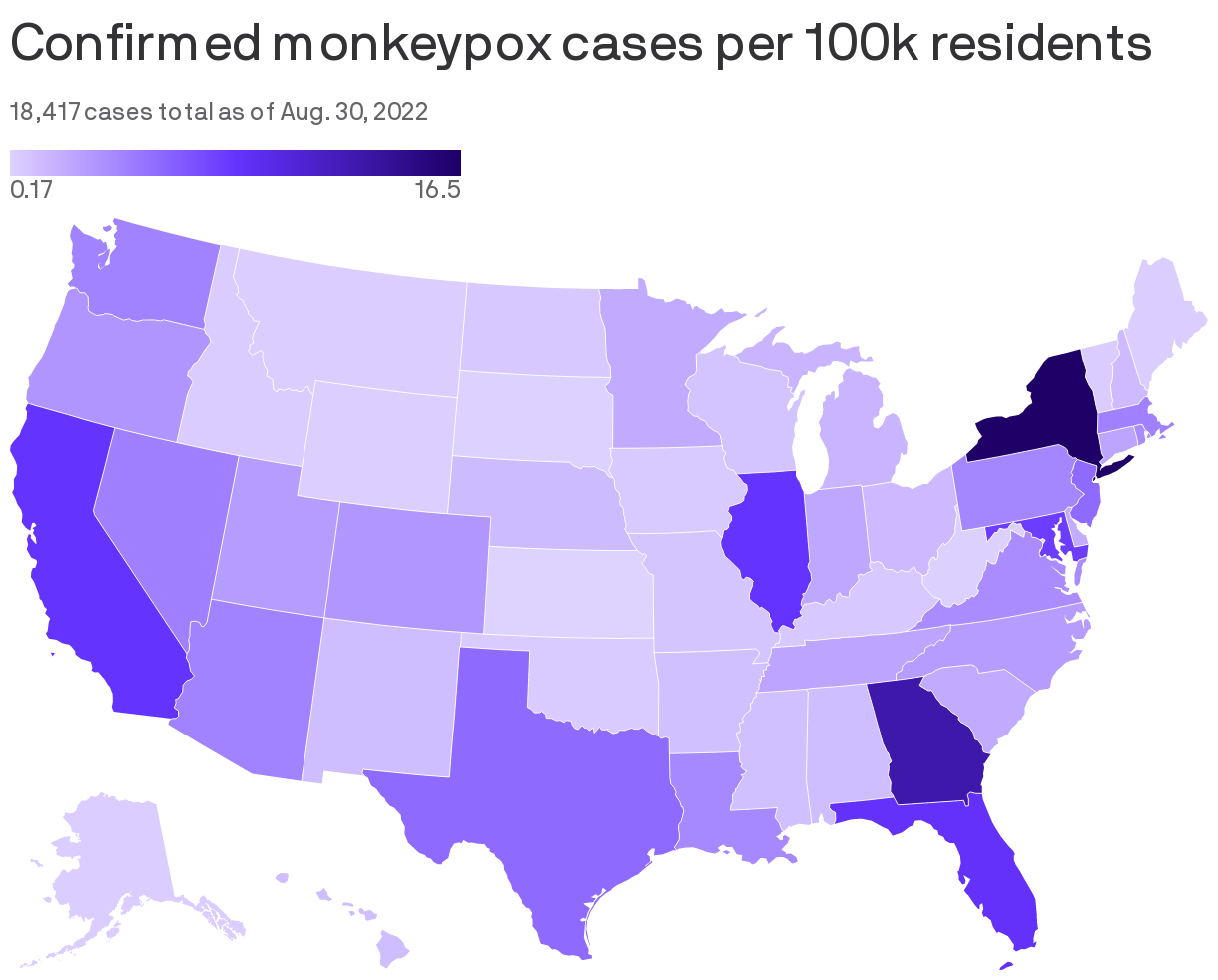
Monkeypox case rates appear to have plateaued nationwide, though there were 677 new cases reported after Labor Day weekend, per CDC data.
- So far, more than 21,000 cases of monkeypox have been confirmed nationwide.
By the numbers: The AIDS Clinical Trial Group is running the trial at several locations nationwide, enrolling more than 500 people who test positive for monkeypox, including high-risk individuals and pregnant people.
- Among 331 patients with monkeypox, 23 were hospitalized, the report shows.
- The majority of patients treated with tecovirimat recovered without symptoms by the time of their last assessment with a provider.
- Most patients began to see their symptoms improve about three days after taking the medication.
The bottom line: Despite some groups advocating for broader access to tecovirimat, federal officials and agencies say they need clinical trial data first.
