Apr 14, 2022 - Health
Pfizer says booster hiked immune response in children ages 5 to 11
Add Axios as your preferred source to
see more of our stories on Google.
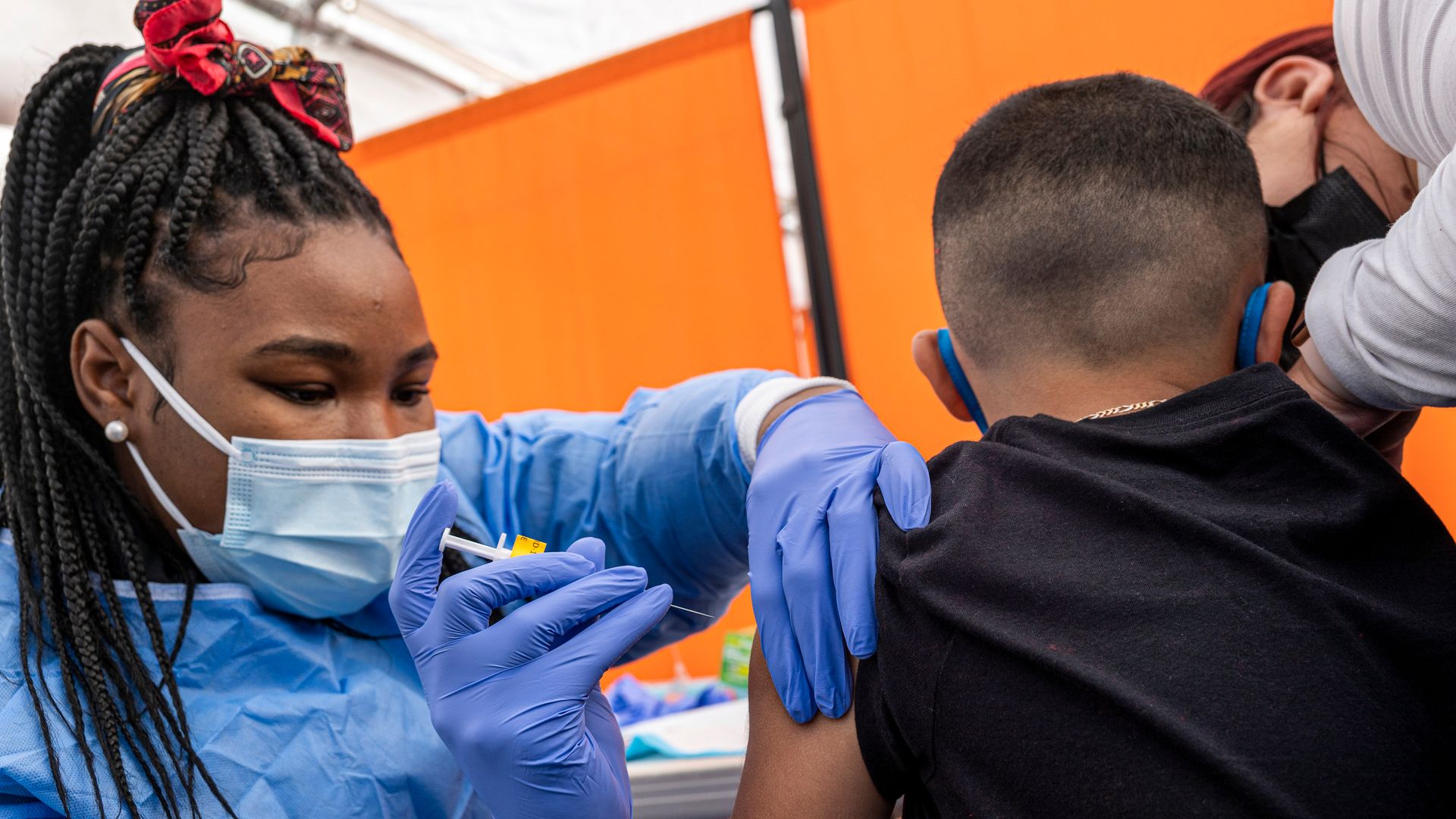
A health care worker administering a Pfizer coronavirus vaccine to a child at a facility in San Francisco in January. Photo: David Paul Morris/Bloomberg via Getty Images
