Controversial Alzheimer's drug approval ignites FDA reform debate
Add Axios as your preferred source to
see more of our stories on Google.
The FDA's conditional approval of a controversial Alzheimer's drug last year has sparked heightened scrutiny and an attempted overhaul of a popular regulatory pathway used to fast-track cancer drugs and certain other treatments.
Why it matters: Accelerated approval allows patients to access new drugs deemed to meet unfilled needs much faster than if the drug went through the regular approval process. But critics say that more needs to be done to prove these drugs actually work in the real world, which could have big implications for the pharmaceutical industry.
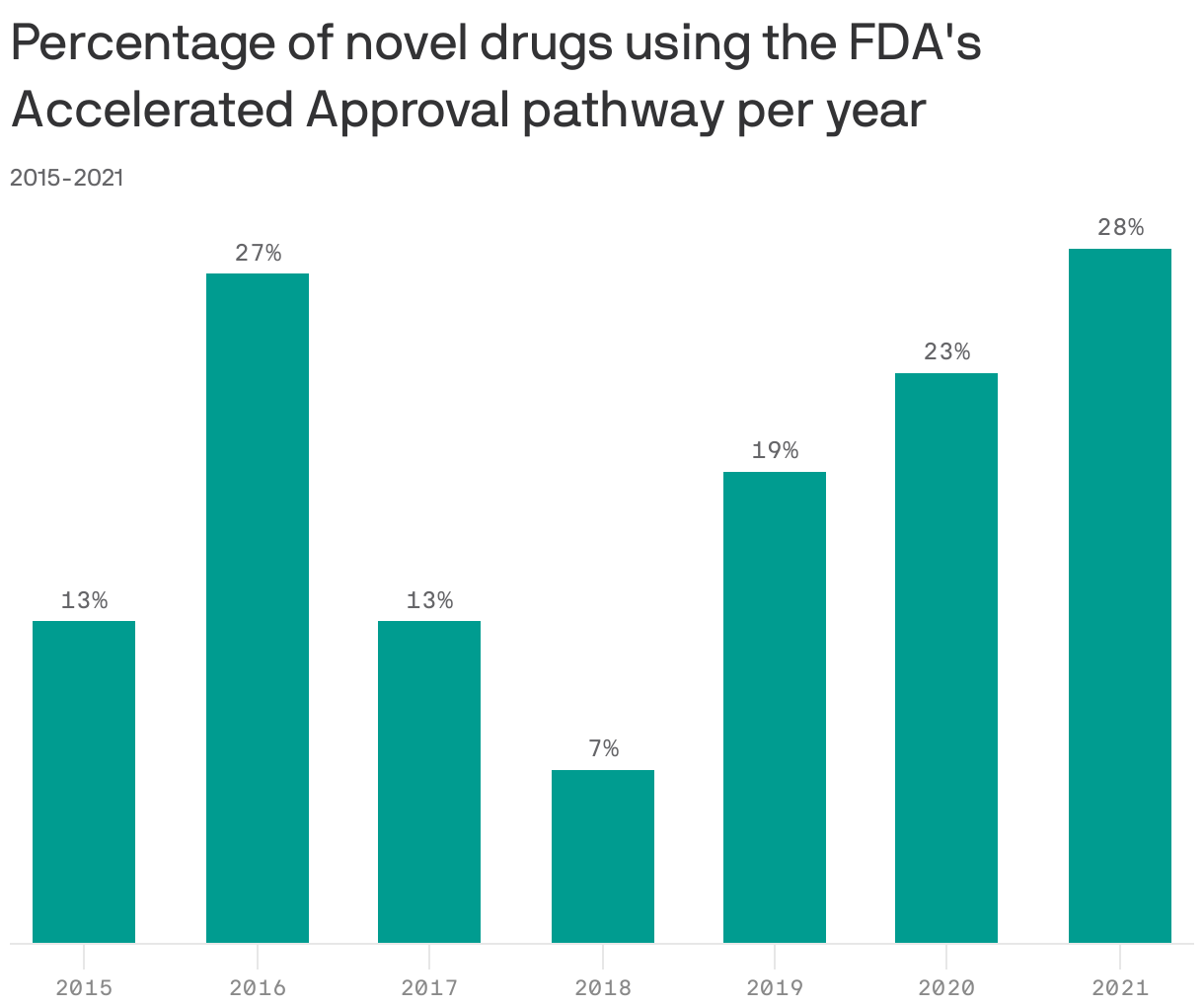
State of play: Last year, 14 of the 50 new drugs approved by the FDA — including Biogen's Alzheimer's drug, Aduhelm — went through the accelerated approval pathway.
- The pathway speeds up the drugs' market entrance by allowing the approval to be based on surrogate endpoints — or variables that suggest the drug likely works — rather than real-world clinical results.
- But the Aduhelm approval came amid significant doubts that the surrogate endpoint used for its approval proved the drug was effective, and the FDA's own advisory board had recommended against its approval.
- Drugmakers are required to perform confirmatory trials in the real world after receiving an accelerated approval. But in the case of Aduhelm, the FDA gave Biogen around nine years to conduct its trial, a period many experts thought ridiculously long to let an unproven drug remain on the market without confirmation that it works.
The big picture: Although most experts and policymakers laud the pathway as a way to deliver lifesaving medicines to patients in a more timely manner, Aduhelm's approval has raised questions about whether the program is being misused.
- The controversy has put a spotlight on what critics say is a system that makes it too easy for drug companies to delay confirmatory trials or in which the FDA doesn't enforce the rules around such trials strongly enough. The critics say the agency also may not have enough power to pull those drugs that fail the follow-up trials.
- Now, influential members of Congress have picked up on these criticisms, the HHS internal watchdog is reviewing the program and the agency itself may even be stepping up its enforcement efforts.
- “This has led to unintended consequences and scrutiny for an expedited program that was generally appropriately utilized," said former FDA deputy commissioner Anand Shah.
Driving the news: The powerful House Energy and Commerce Committee is considering legislation sponsored by Chairman Frank Pallone that would reform that pathway as part of a larger medical innovation package.
- "Under the current system, some sponsors have failed to conduct trials in a timely manner," Pallone wrote before a hearing on the bill last week. "Patients deserve to know that the drugs they are taking are safe and effective."
- Some lawmakers, including Senate Finance Committee Chairman Ron Wyden, have pointed out that the agency could use its existing authorities more vigorously without legislative changes.
In a letter replying to Wyden's concerns about accelerated approval, recently-confirmed FDA commissioner Robert Califf said that ensuring follow-up studies are done in a timely manner will be a "high priority."
- But even before his confirmation, the agency withdrew several accelerated approvals for cancer drugs last year and more have been withdrawn already this year.
- “I do think FDA is trying to enforce these requirements in a way that it hasn’t been previously, but also that they may be starting to run into some of the limits of their legal authority," said Rachel Sachs, a law professor at Washington University.
- For example, even though the FDA says a prescription hormone called Makenna — which entered the market through accelerated approval — has failed to prove that it works, the drug is still on the market, the LA Times recently reported.
What they're saying: In written testimony to the E&C committee last week, a PhRMA official said the accelerated approval pathway "is a critical tool for patients and regulators, and the biopharmaceutical industry continues to support the pathway in its current form."
- "Because most drugs do convert from accelerated to traditional approval within the five-year timeline, for most companies there would be no significant changes to their existing practices," Sachs said. "But companies in general are opposed to any efforts to constrain their ability to get these drugs to market and to sell them.”
The other side: Some critics are more cynical about the industry's support of the status quo.
- "They've figured out it's a lower bar to get accelerated approval. They can then dictate the trial, and something can get on the market quite rapidly and not get pulled off the market," said Zeke Emanuel, a professor of medical ethics and health policy at the University of Pennsylvania.
- "Their business model would have to change, because they'd actually have to finish these studies," he added.
