Updated Feb 1, 2022 - Health
Pfizer asks FDA to authorize COVID vaccine for kids under 5
Add Axios as your preferred source to
see more of our stories on Google.
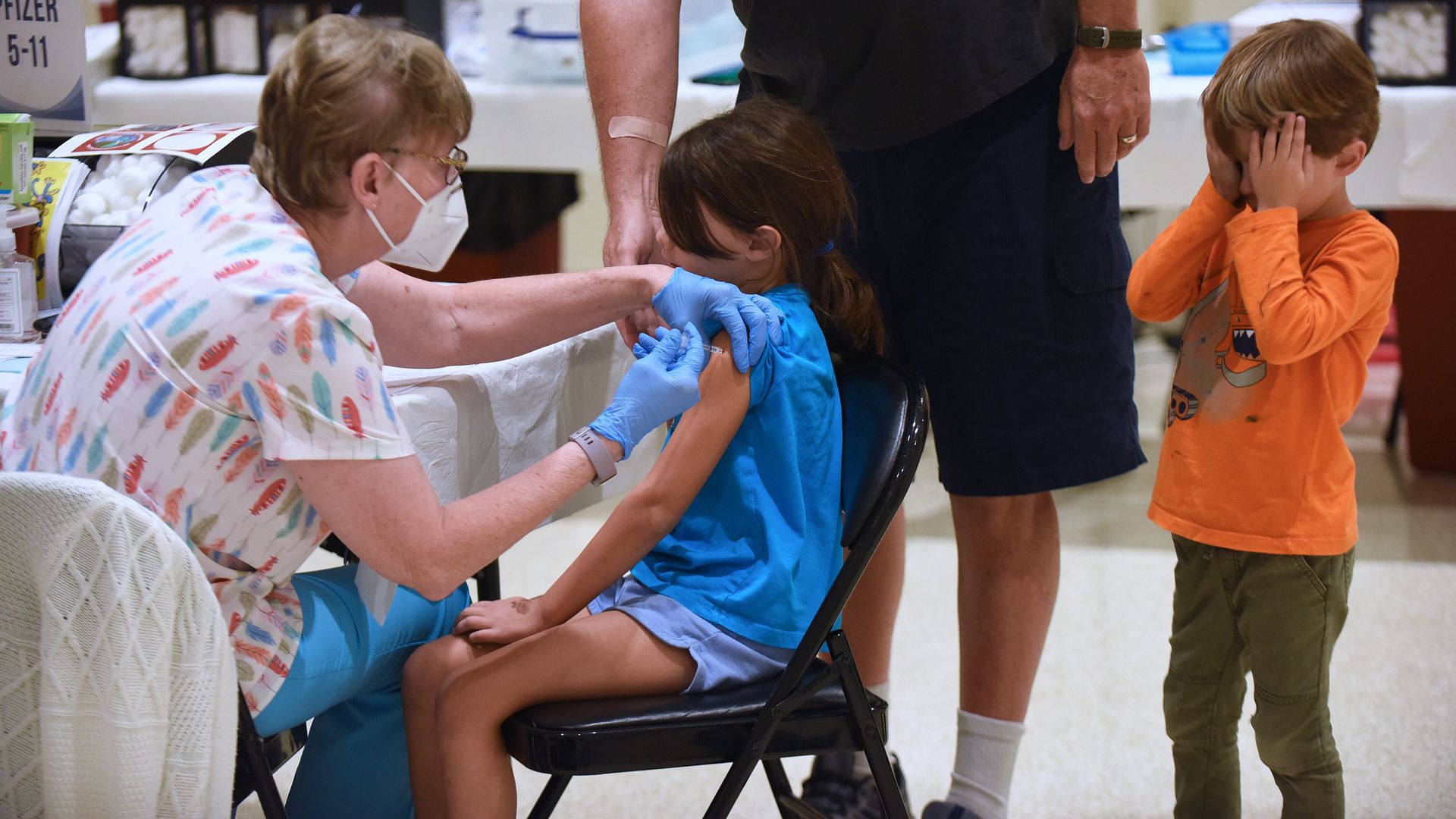
A nurse gives a little girl a shot of the Pfizer COVID-19 vaccine while her brother covers his eyes at a vaccination site in Florida. Photo: Paul Hennessy/SOPA Images/LightRocket via Getty Images
