Dec 10, 2020 - Health
FDA advisory panel endorses Pfizer coronavirus vaccine for emergency use
Add Axios as your preferred source to
see more of our stories on Google.
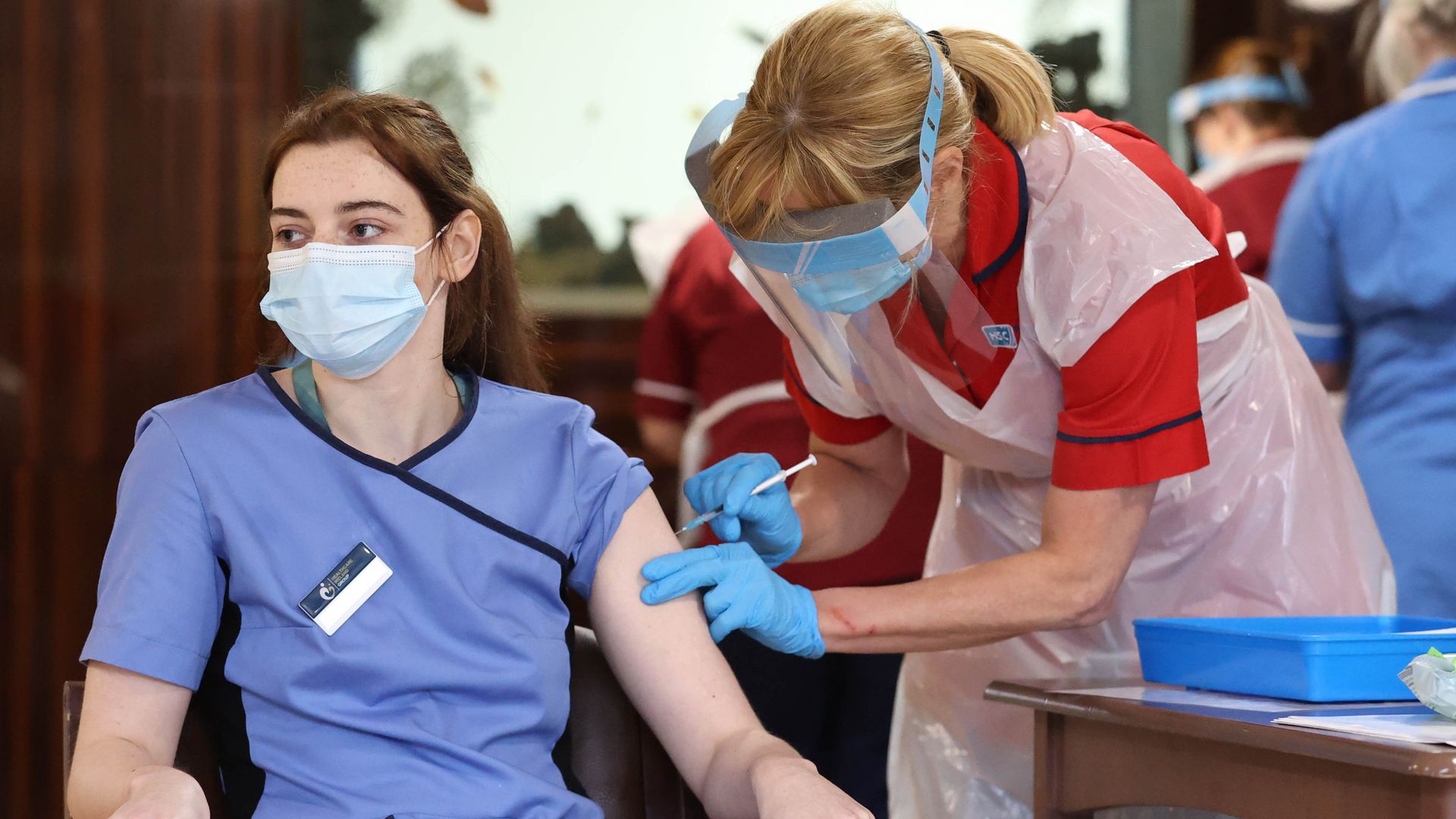
A care home staff receiving a dose of the Pfizer/BioNtech coronavirus vaccine in Belfast on Dec. 9. Photo: Liam McBurney/PA Images via Getty Images
