An "operating system for the brain" gets FDA approval
Add Axios as your preferred source to
see more of our stories on Google.
Courtesy of MindMaze
Combining virtual reality and neuroscience may help a stroke patient's brain recover more quickly.
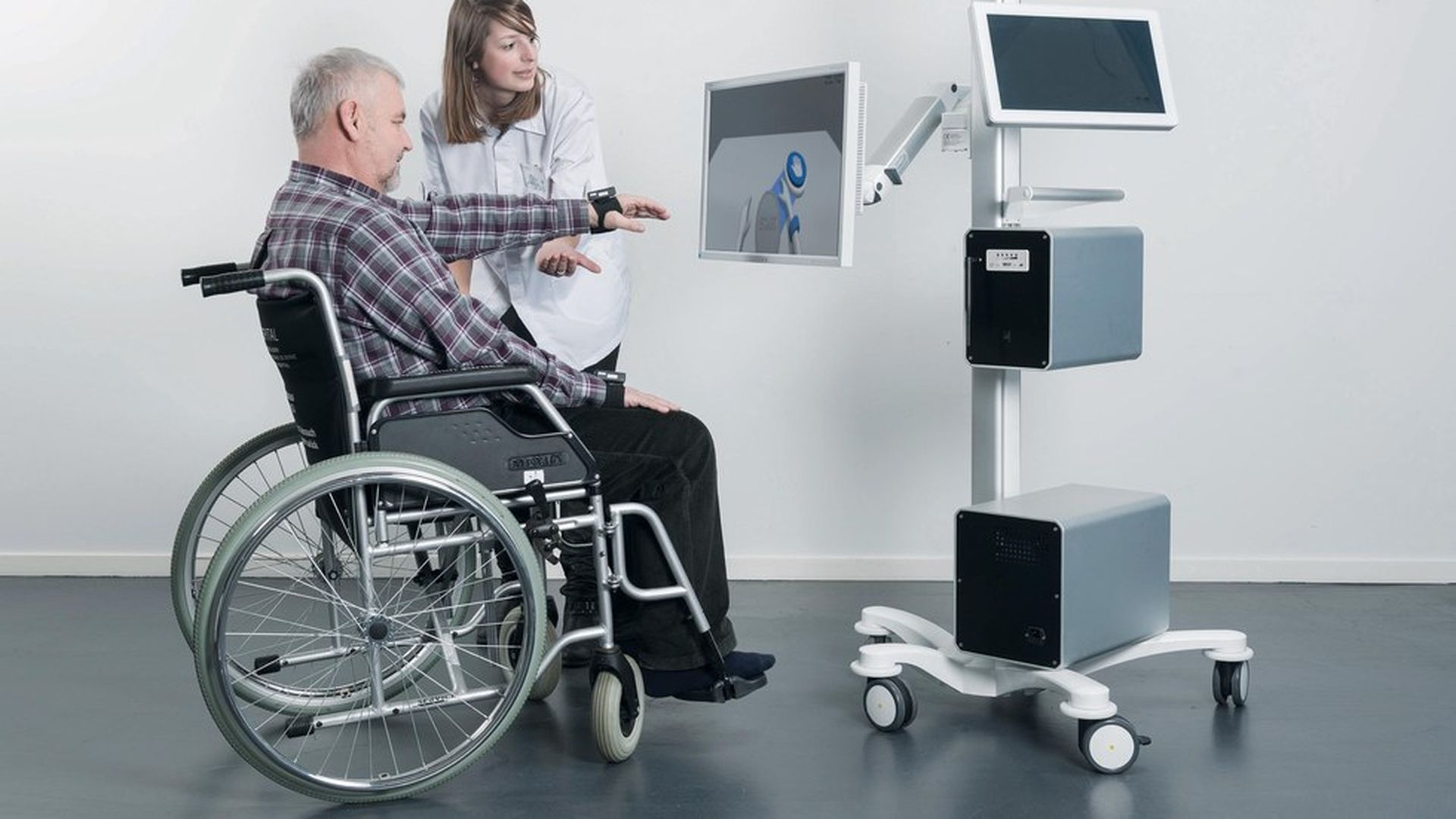
That's according to MindMaze, a Swiss company (with U.S. headquarters in San Francisco) that just received FDA approval to bring its virtual reality platform to the U.S. market, after already selling it in Europe. The "neurorehabilitation" platform, called MindMotion Pro, uses 3D motion tracking cameras to coordinate movement and brain function and then analyzes that data to tailor therapy, CEO and co-founder Tej Tadi told Axios.
Why it matters: A growing number of virtual reality use cases are in healthcare settings, for both medical training and assisting with therapy. Now that it is FDA-approved — a big win for any startup— MindMaze's virtual reality device will be among the first to be used in U.S. hospitals to assist with therapy after brain injury.
How it works: Using sensors and tracking cameras, MindMotion Pro maps a patient's movements of arms or fingers on a 3D avatar seen on the video-game-like platform. Even if a patient can't actually go through the motions, imagining that they are doing so through the avatar helps activate damaged areas of the brain trigger new neural activity.
Tadi calls it "an operating system for the brain," which can repair a damaged brain and enhance learning for a healthy one. Early partners include Stanford and University of California San Francisco.
It's not cheap: A hospital-grade device runs about $80,000, Tadi said.
