Mini lab-created organs successfully check cancer treatments
Add Axios as your preferred source to
see more of our stories on Google.
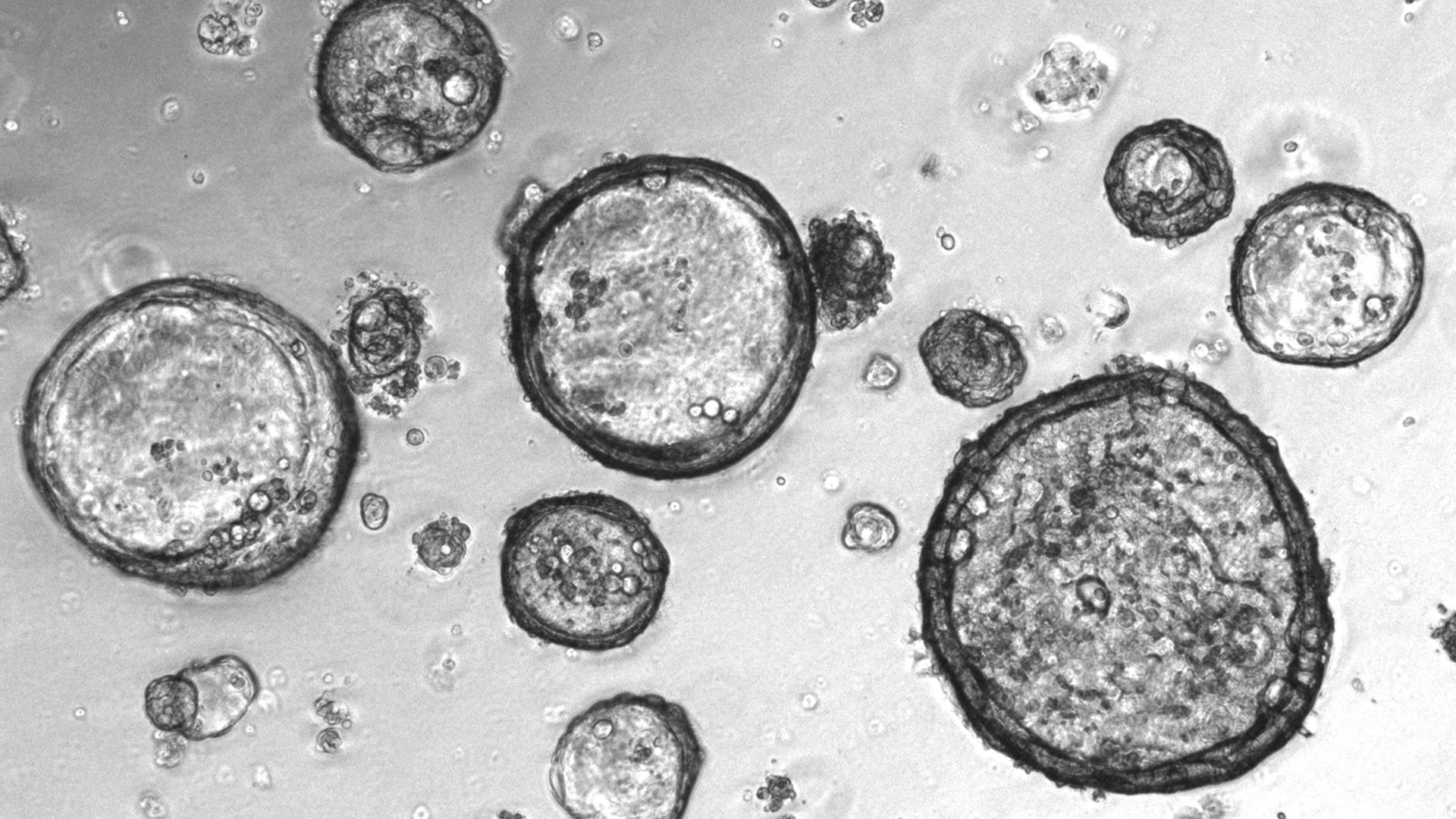
Microscopic images of lab-grown organoids from a patient with metastatic gastroesophageal cancer. Photo: George Vlachogiannis / Institute of Cancer Research at the Royal Cancer Hospital
Scientists used lab-grown cells from an individual's cancer tumor to study their response to different drugs, according to a new study published in Science Thursday.
Why it matters: These cells, called organoids, bring us a step toward honing precision medicine for patients with aggressive, metastatic gastrointestinal cancers.
"It could allow clinicians not only to test known anticancer drugs but also other drugs that are not being used in cancer but that could be re-purposed for that particular patient."— Meritxell Huch of the University of Cambridge's Gurdon Institute
Hans Clevers, group leader at the Hubrecht Institute for Developmental Biology and Stem Cell Research, who was not part of this study but helped create the organoid technique used in this trial, tells Axios:
"It proves beyond a doubt that cancer organoids can be used to predict drug response in a personalized fashion. We have given some anecdotal evidence earlier, but nothing to the level of this paper."
What they did during the 3-year trial:
- The researchers took 110 fresh tumor biopsies from 71 gastrointestinal patients and used them to create patient-derived organoids.
- They compared the organoids with the original tumors to check they were similar.
- They investigated the robustness of organoids as a screening platform for drug discoveries, and compared head-to-head the organoid response with the patient's response in the clinic.
- It took roughly 6–8 weeks from biopsy to obtain the drug results.
What they found: The team found 88% positive predictive value (ability to identify responsive drugs) and 100% negative predictive value (ability to identify non-responsive drugs) in forecasting response to targeted agents or chemotherapy in patients.
"The organoids could recapitulate (ex vivo) patients' treatment responses to all currently approved anti-cancer treatment regimens used for metastatic colorectal cancers. The fact that these observations were not restricted to certain treatment was suggestive of the robustness of our experimental approach, but more importantly of the power this system may have in predicting clinical responses, which can have major clinical implications for personalized medicine."— George Vlachogiannis, study author from the Institute of Cancer Research in London
More: They found if a sample doesn't have a lot of tumor cells — which means it's hard to get good results via genomic sequencing — organoids can still be grown to help make a diagnosis.
"This is per se very important as it allows performing an accurate genetic analysis of the tumor even in those cases where this is not possible from the tumor biopsy directly," Huch tells Axios.
Limitations: Emile Voest, who was not part of this study, says more research on the variety of drugs and on a greater number of patients is needed before it can be decided if there is potential clinical applications.
"This is the next challenge for the field: are tumor organoids able to be used as a predictive test to select or avoid a treatment."— Voest, medical director of the executive board of the Netherlands Cancer Institute
Vlachogiannis agrees and adds:
"One needs to remember that cancer is a very complex disease, and that various tumor lesions within the same patient respond differently to treatment. ... Establishing organoid cultures from circulating tumor cells may be a better way to capture cancer heterogeneity within the patient...and efforts should be put in that direction as well."
