Axios Vitals

February 10, 2026
We've got news. Today's newsletter is 911 words, a 3.5-minute read.
1 big thing: Exclusive — House panel subpoenas 8 ACA plans
House Judiciary Committee Republicans have subpoenaed eight Affordable Care Act health insurers for documents as part of a widening investigation of potential fraud surrounding the use of premium subsidies, Axios has learned.
Why it matters: Enhanced premium tax credits for ACA coverage expired on Jan. 1. But Republicans are pressing forward with their probe of the Obamacare market — and putting heat on the entire health insurance industry.
State of play: House Judiciary Chair Jim Jordan (R-Ohio) issued subpoenas yesterday to get health insurers to send lawmakers more information on their subsidized ACA enrollees and discussions on subsidy-related fraud, the committee told Axios.
- The companies are Elevance, CVS, Centene, GuideWell, Oscar Health, Kaiser Permanente, Health Care Service Corporation and Blue Shield of California.
- Each must respond by Feb. 23, according to subpoenas viewed by Axios.
Jordan asked the insurers for documents and information in December after federal auditors showed they'd obtained subsidized coverage for almost two dozen individuals who didn't exist and identified other fraud risks in the system.
- The Judiciary Committee is now looking into whether legislative changes are needed to address ACA fraud, per the Monday letters.
- "The documents requested by the Committee will inform the House's consideration of these potential legislative reforms," they state.
Zoom in: The subpoenas ask for documents showing the number of enrollees at each organization receiving ACA subsidies and how much money it received from the subsidies between 2020 and 2025, according to documents viewed by Axios.
- They also request information on enrollees with subsidies who did not utilize any benefits in a given year, as well as the total amount each insurer pays brokers and agents for work in ACA markets.
Catch up quick: The subpoenas hit as insurers are increasingly under fire from critics in Congress and the Trump administration.
2. New controls on Obamacare planned for 2027
The administration wants to tighten marketing standards and add income verification policies for ACA marketplace plans starting in 2027, according to a proposal released yesterday.
Why it matters: While the changes could reduce fraud and lower premiums, they could make coverage harder to obtain for some low-income enrollees and steer more consumers toward high-deductible plans.
State of play: CMS would make enrollees submit documents to verify their income when they have low earnings, or when tax data isn't available.
- Federal administrators also outlined prohibited conduct by agents and brokers, including falsely claiming that consumers could qualify for "zero-dollar" insurance and using AI to create fake endorsements.
The intrigue: CMS wants to let plans without any provider networks be sold on the Obamacare marketplaces, if they can ensure access to enough providers that accept their benefit amount as payment.
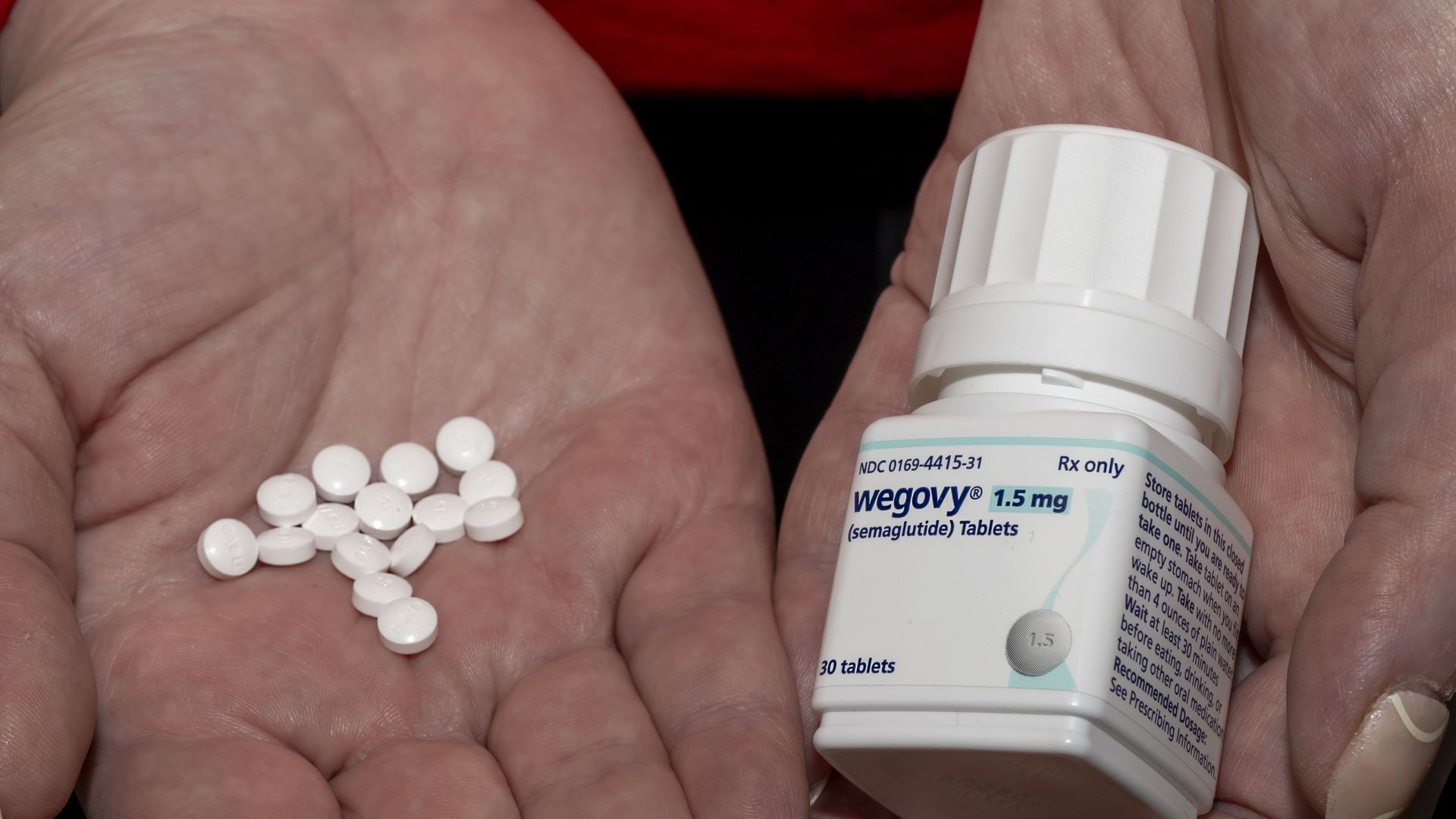
3. FDA: Novo made misleading claims on GLP-1 pill
The FDA accused Novo Nordisk of airing false and misleading claims about its new Wegovy pill in a TV advertisement and requested the drugmaker take action, including potentially pulling the ad and others with similar claims.
Why it matters: The warning letter sent on Feb. 5 complicates the rollout of the oral version of the blockbuster weight-loss drug that Novo Nordisk is hoping will help it regain dominance in the anti-obesity market.
Driving the news: FDA said the ad's claims "live lighter" and "a way forward" misleadingly imply the pill will deliver additional weight loss compared with other currently approved GLP-1 treatments, which hasn't been proven.
- They also promise benefits beyond physical weight loss, "positioning the drug as a solution to broader life challenges rather than a treatment for a specific condition," the letter states.
- FDA also said the ad additionally didn't present risk information using both audio and text, as is required.
- Novo Nordisk acknowledged receiving the letter and said it's responding.
Context: The Trump administration is getting aggressive policing advertising around GLP-1 weight-loss drugs amid surging demand.
- Last week, HHS asked for an investigation of whether the telehealth brand Hims & Hers violated the Food, Drug and Cosmetic Act by planning to sell a cheaper, compounded version of the Wegovy pill.
- The company backed off those plans on Saturday.
4. First look: New York joins WHO network
New York today became the latest state to join a World Health Organization network that monitors and responds to disease outbreaks.
The big picture: The move is part of blue states' efforts to push back against Trump administration policies that also featured the creation of a 14-state public health alliance last October.
- California and Illinois opted into the WHO network earlier this year after President Trump formally withdrew the U.S. from the global health agency.
Driving the news: New York Democratic Gov. Kathy Hochul's office, in a statement first shared with Axios, said it was sharing expertise, laboratories and a highly skilled workforce to detect and respond to outbreaks while helping prevent health threats from reaching the state.
- The New York City Health Department announced last week that it had joined the WHO network.
- The network includes more than 300 technical institutions worldwide.
5. Catch up quick
🏥 A strike at Kaiser Permanente entered its third week with thousands more workers joining to protest what they argue are unsafe staffing levels. (Healthcare Dive)
👀 The latest batch of documents related to Jeffrey Epstein show scientific and medical researchers consulting the financier and sex offender on publications, visas and more. (Nature)
🤖 As AI enters the operating room, reports arise of botched surgeries and misidentified body parts. (Reuters)
Thanks for reading Axios Vitals, and to editors Adriel Bettelheim and David Nather and copy editor Matt Piper. Please ask your friends and colleagues to sign up.
Sign up for Axios Vitals