Axios Vitals

March 31, 2022
Good morning, Vitals readers. Today's newsletter is 1,054 words or a 4-minute read.
1 big thing: Dems might use legal muscle on drug patents...
Illustration: Aïda Amer/Axios
The decades-long debate over whether the federal government can and should use its legal muscle to lower prescription drug prices may be reaching an inflection point, especially with President Biden's drug pricing agenda stalled on Capitol Hill, Axios' Caitlin Owens writes.
Why it matters: Democrats would love to show voters they've acted to lower drug prices before the November midterms, and progressive groups are urging them on.
- But taking on pharma through executive action risks uncertain legal outcomes, and opponents say that it could have a chilling effect on innovation.
State of play: Democrats' dreams of sweeping drug pricing proposals — including Medicare negotiations, which is hugely popular with their base — are being dashed in the Senate with the rest of Biden's domestic agenda.
- If the stalemate continues, that would pressure the administration to find ways to lower prices administratively. In a drug pricing plan that HHS released in September, the agency said using legal authorities to circumvent drugmakers' patents on medicines developed with the government's help is still on the table.
The intrigue: A test case of how far the administration is willing to go is already teed up.
- The NIH is reviewing a request to lower the price of Astellas and Pfizer's prostate cancer drug Xtandi by using "march-in" rights, which allow federal agencies to remove exclusive patent rights on drugs that were developed using federal funding. The drug has an annual cost of about $160,000.
- The primary legal debate is over whether exorbitantly high prices count as justification for the government taking control of rights. The NIH has previously said that it doesn't believe so.
- But the retirement of former NIH director Francis Collins, who was opposed to using march-in rights to lower prices, may create an opportunity for change.
2. ...or they might go with a win on insulin caps
Illustration: Sarah Grillo/Axios
Congress is pressing ahead with plans to limit the cost of insulin to $35 a month — an effort that could be the closest Democrats get to making good on campaign pledges (see above) to rein in drug costs, Axios Adriel Bettelheim writes.
Why it matters: But while capping out-of-pocket costs for insulin might be a political winner in an election year, it could wind up costing the government billions in lost revenue and swelling the deficit.
Zoom in: Insulin is a tempting target. Per-unit U.S. prices for the life-saving medication in the U.S. are seven times those in comparable countries, per the Peterson-KFF Health System Tracker.
- But legislation like a House bill from Rep. Angie Craig (D-Minn.) that'll be debated on the floor today wouldn't address the core problem of rising prices for insulin. It would instead shift more of the cost onto health insurers and employers and result in higher premiums, according to experts.
Go deeper: Congressional scorekeepers say the Craig plan would lead the government to lose almost $4.8 billion in revenue over a decade while spending would rise $6.6 billion over that period.
- Yes, but: Craig's bill and similar measures proposed by Sens. Raphael Warnock (D-Ga.), John Kennedy (R-La.) and others would still save money for people with diabetes and keep insured people from spending thousands of dollars.
Look ahead: Senate Health, Education, Labor and Pensions Committee Chair Patty Murray (D-Wash.) and Warnock plan to rally support for the $35 cap at a virtual event tomorrow.
3. New COVID cases in U.S. leveling off
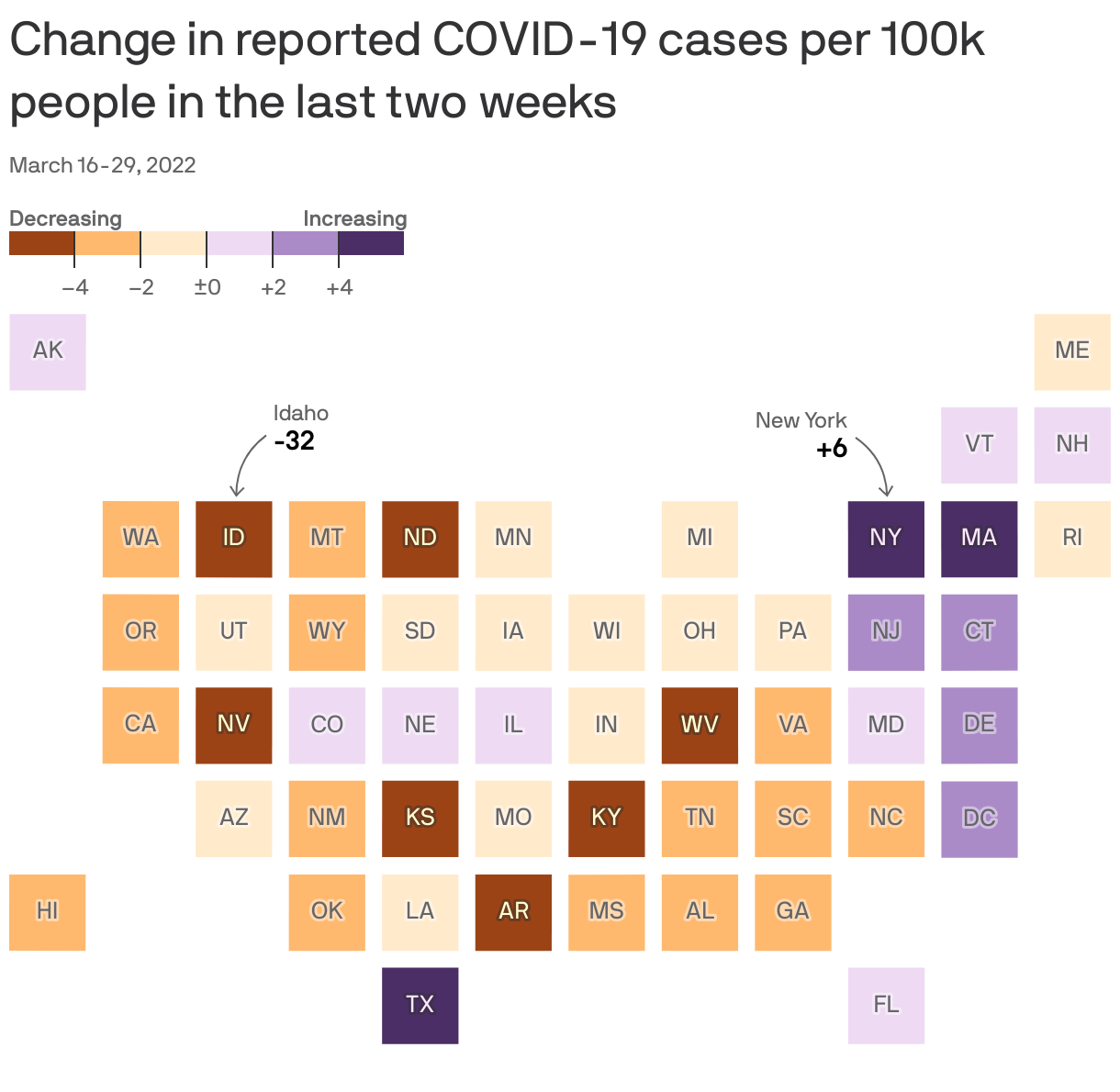
After plummeting for several weeks, the number of new COVID cases in the U.S. has largely leveled off, Axios' Kavya Beheraj and I report.
Driving the news: The CDC confirmed this week the Omicron subvariant BA.2, which has been driving surges of COVID elsewhere around the world, has become the dominant strain circulating in the U.S.
By the numbers: The U.S. is now averaging roughly 29,253 new COVID cases per day, a 6% drop from the rolling average of about 31,000 cases two weeks ago (and about even with new cases just a week ago).
- Kansas and Louisiana had the lowest case rates with two new cases per 100,000 people. Alaska has the highest case rate with 29 new cases per 100,000.
- Idaho saw the greatest drop in new cases over the past two weeks with rates dropping from 38 cases to six cases per 100,000 people over the past two weeks.
- While cases are still comparatively low, three states — New York, Massachusetts and Texas — reported some of the biggest increases in new cases.
Of note: There were just more than 700 daily deaths on average from COVID, down from more than 1,200 two weeks ago.
4. Convalescent plasma works, study finds
Convalescent plasma can work as well as other COVID treatments authorized for early use, according to a study published Wednesday in the New England Journal of Medicine.
Driving the news: The results of this study were first reported in December before it was peer-reviewed. It was also before researchers had a chance to consider Omicron.
- Since then, researchers have continued collecting data that has reaffirmed the ongoing effectiveness of convalescent plasma when it has high antibody levels and is administered early in the course of COVID-19 illness, even against Omicron.
- "Omicron showed us there is a place for convalescent plasma and it is being used for immunosuppressed patients where the monoclonals were defeated by the variants," said David Sullivan, a professor at the Johns Hopkins Bloomberg School of Public Health and study co-lead author.
Details: In the study led by researchers at Johns Hopkins Medicine and the Johns Hopkins Bloomberg School of Public Health, patients were given either one dose of antibody-rich convalescent plasma, or placebo plasma.
- They found antibody-rich convalescent plasma reduced the need for hospitalization for more than half of the study's largely unvaccinated patients when delivered within nine days of testing positive.
- If convalescent plasma was given within five days after diagnosis, the effectiveness at reducing hospitalization is about 80%, the researchers said.
Related: Ivermectin does not reduce risk of COVID hospitalization, large study finds
5. Catch up quick
President Biden receives a fourth dose of the Pfizer/BioNTech COVID-19 vaccine on Wednesday. Photo: Anna Moneymaker/Getty Images
- President Biden made another appeal for Congress to pass COVID funding that would help pay for, among other things, fourth doses of vaccines for Americans. (ABC News)
- In his first interview as CEO of Johnson & Johnson, Joaquin Duato said there will be more innovation in health care over the next 10 years than in the past century. (CNBC)
- HHS will fly a trans pride flag in honor of Transgender Day of Visibility, becoming the first federal agency to do so. (Axios)
Sign up for Axios Vitals

Healthcare policy and business analysis from Tina Reed, Maya Goldman, and Caitlin Owens.