Axios Vitals

November 10, 2017
Good morning ... And happy Veterans Day.
Deal would keep Pentagon out of product approvals
Lawmakers are approaching a bipartisan deal to resolve the ongoing dispute over whether to let the Defense Department approve medical products in an emergency, Sen. Patty Murray told me yesterday.
The issue: A provision in this year's defense authorization bill would permit the Defense Department to allow the use of certain medical products — such as freeze-dried plasma — on the battlefield, even if they haven't been approved by the Food and Drug Administration.
- That touched off a bipartisan rebellion from members of the Senate HELP Committee, who feared the provision would undermine the FDA's authority and the rigorously controlled process it uses to evaluate new treatments.
The deal: Product reviews remain solely within the FDA's jurisdiction, but would give the Pentagon new powers to get the FDA to expedite those reviews, according to the agreement reached between Murray and Sen. Lamar Alexander.
- Under the agreement, if the defense secretary asks for it, the FDA "shall take action to expedite the development and review of an applicable application" for relevant medical products.
- Officials from the FDA would have to meet quarterly with Pentagon officials to figure out the military's top priorities (the bill specifically mentions freeze-dried plasma as one of those priorities).
- The FDA already has the power to allow the use of unapproved drugs in emergency situations, as it did with experimental Ebola treatments in 2014, and the Pentagon can already have a say in that process. This agreement would tweak those rules to give the Pentagon some more flexibility.
What's next? A Senate committee aide told me this is the deal that's likely to become final. While it's not clear how this would get passed — on its own or as an attachment to something else— Congress does not seem interested in slowing down the defense bill to address this.
ACA enrollment off to a strong start
Who saw this coming? ACA enrollment is off to a stronger start this year than last year. Roughly 600,000 people selected plans through HealthCare.gov in the first four days of open enrollment, which breaks down to about 150,000 per day — compared with 84,000 per day in the first two weeks of the last sign-up period.
Key stat: The share of new and returning customers is about the same this year as it was at the beginning of last year's enrollment period.
- When we first started hearing rumblings that enrollment was up, some experts wondered if that spike was just driven by a lot of people renewing their coverage. Turns out, it wasn't.
Yes, but: This is a surprisingly strong start, but we'll have to see how the rest of the sign-up period plays out.
- It's hard to know for sure who's signing up, but it stands to reason that sicker people will be more eager to get covered, while healthier people are more likely to come in toward the final deadline.
- Healthier people are also the ones most likely to need some prodding to sign up, so the Trump administration's cuts to outreach programs could still make a difference.
- These numbers are subject to adjustment. Some people who signed up for plans won't go on to pay their premiums, and won't actually be covered. That's normal.
Senate's tax bill could be bad news for pharma
Pharmaceutical companies could see their tax bills rise sharply under the Senate's proposed tax overhaul. The bill would impose a 12.5% tax on the income U.S. companies earn from intellectual property, irrespective of whether that intellectual property is housed here or abroad.
As the Wall Street Journal explains, that provision appears to be aimed at least partially at drug makers, who have taken to parking their patents overseas in an effort to avoid paying U.S. taxes.
Here's what else you need to know about the health care components of the new tax plans Republicans released yesterday.
- The Senate's plan does not include repealing the ACA's individual mandate.
- Ways and Means chairman Kevin Brady indicated that the House is unlikely to add individual-mandate repeal to its tax bill, Roll Call reports.
- Unlike the House bill, the Senate's proposal would not repeal existing tax deductions for especially high medical expenses or for adoption.
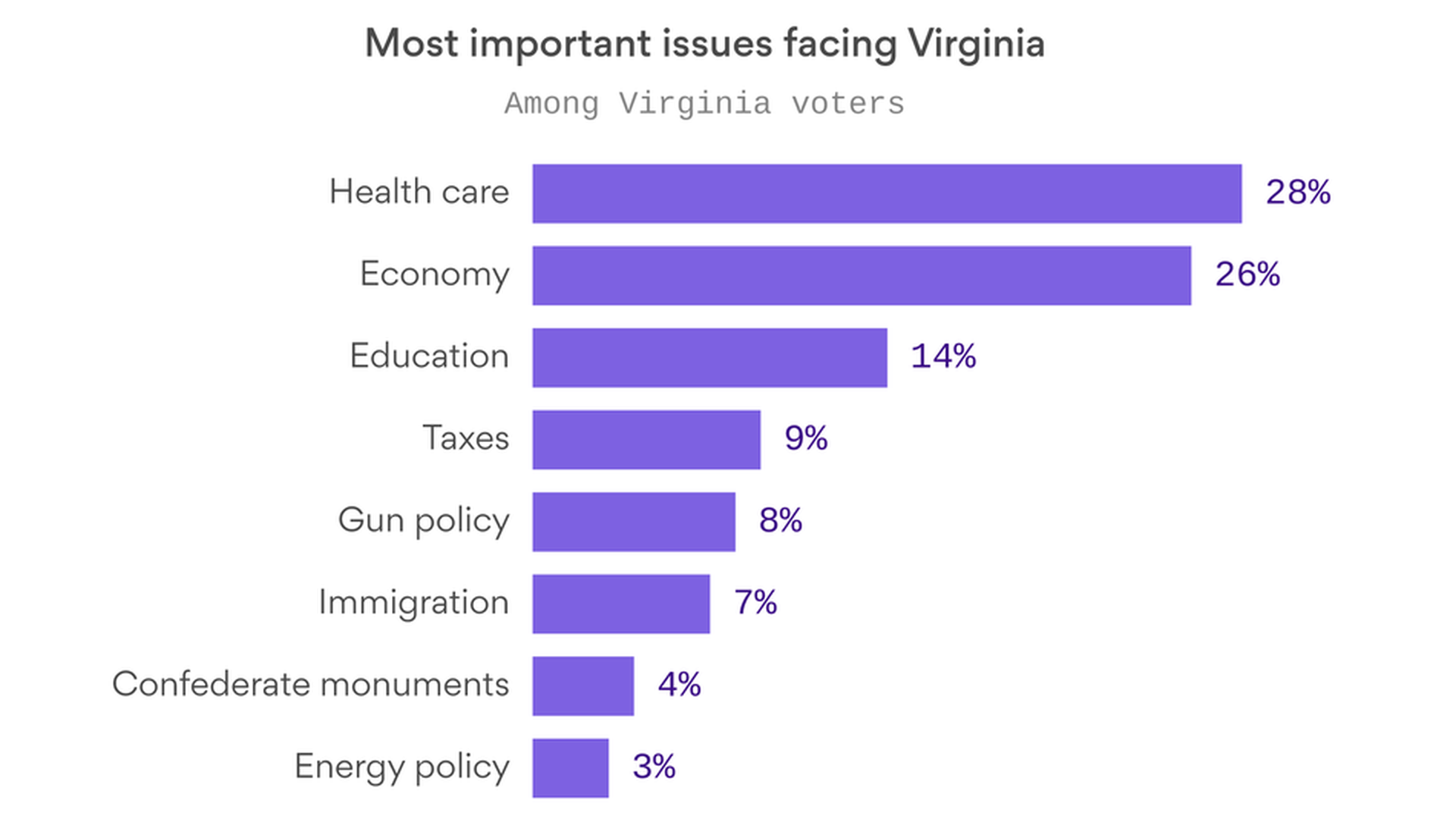
What Tuesday's elections mean for health care
When voters rank health care as a top issue in an election — as they did in Virginia's gubernatorial election, according to exit polls — it does not necessarily mean health care drove their vote, Kaiser Family Foundation president Drew Altman writes in his latest Axios column.
But between voters' avowed health care focus in Virginia, and their vote to expand Medicaid in Maine, Tuesday's races still tell us a lot about where the politics of health care are headed.
The impact: Expanding Medicaid could be a winner in other states, especially with the federal government picking up 90% of the costs and the Trump administration ready to let red states put a conservative stamp on their programs. Medicaid is not Social Security or Medicare yet, but politically it is a lot closer than Republicans may realize, Altman writes.
And the prominence of health care in the Virginia election could throw a scare into moderate Republicans about continuing to pursue ACA repeal.
Things aren't looking so hot for Soon-Shiong
If you've been seeing a lot of red ink lately, it's likely coming from NantHealth, the cancer testing company founded by the politically connected billionaire and doctor Patrick Soon-Shiong.
What's happening: My Axios colleague Bob Herman reports that NantHealth, which already laid off 300 employees over the summer, lost more than $42 million in the third quarter and has burned through more than half of its cash since the start of the year.
Why it matters: Soon-Shiong has the deep pockets to keep this ship afloat (NantHealth has lost $338 million since the start of 2016). But there's no evidence the company's personalized cancer tests are winning over employers, insurers or patients, and it's unclear how much patience investors have left.
Go deeper: STAT and Politico both have deeply reported pieces about Soon-Shiong, and the questionable practices surrounding his company.
The next big thing in cancer treatment
My Axios colleague Eileen O'Reilly, who helps get this newsletter into your inbox every morning, takes a look this morning at one of the next big advances in cancer treatment: blood tests that can monitor the growth of tumors by analyzing DNA shed from tumors into the bloodstream.
Why this is important: Cancers respond differently in each person, and tumor DNA analysis may help to unravel what might work for a particular patient.
- "We can now look not only gene-by-gene but throughout the entire cancer genome in order to figure out which treatments are right for which patient, and why some cancer cells don't respond to therapy. Doing so may inform better ways to treat the entire cancer and to prevent it from coming back," Viktor Adalsteinsson, the author of a new study on this process, told Eileen.
Go deeper: The National Cancer Institute has a thorough explainer of how blood biopsies are used to detect, track and treat cancer.
I want your tips! Also your questions, comments and other feedback: [email protected]
Sign up for Axios Vitals

Healthcare policy and business analysis from Tina Reed, Maya Goldman, and Caitlin Owens.