Axios Vitals

November 03, 2025
🌅 Welcome to Monday! They haven't solved the Obamacare subsidies yet. Today's newsletter is 1,166 words or a 4.5-minute read.
- Just a reminder that you can send us sensitive news tips on Signal at MayaGold.57, TinaReed.202 or Petersu.98.
1 big thing: The rural health money race
There's about to be a lot of competition for the $50 billion rural health fund that was created to minimize the impact of the GOP budget law — and rural providers worry they're going to get squeezed out.
Why it matters: The money could provide a lifeline as rural providers across the country struggle to keep facilities open under the nearly $1 trillion in Medicaid cuts anticipated from the budget bill.
- But many are concerned they could get outhustled, with large academic medical centers, private equity-backed providers, technology companies and startups also pitching states for ways to use the money.
- "I was surprised by how many people have come to the table wanting a piece of the pie," said Terry Scoggin, interim CEO of the Texas Organization of Rural and Community Hospitals — including some "that have not been rural stakeholders in the past."
How it works: Congress laid out a two-step process for awarding the funds. CMS will distribute half of the $50 billion equally to states that apply.
- But states have to compete for slices of the other half. Their applications are due Wednesday.
Writing the applications is just the first part of the funding process. Once states get the money from CMS, they'll have to figure out how to distribute it to providers and organizations.
- Scoggin said he's been impressed with how Texas has worked with rural providers so far to get feedback for the application. But he worries the next step could be where rural providers get left behind.
- "Rural hospitals don't need competitive grants. We don't have money for grant writers and vendors," he said. "We need to make it simple and not who can make the prettiest PowerPoint gets the money."
- "I think the state heard that," he added.
What they're saying: The state applications come first. Geisinger Health, which serves 1.2 million people across largely rural areas of Pennsylvania, told Axios that it wanted more of a say in how the state writes it.
- "We want to help ensure that it's represented in a way that allows Pennsylvania ... the best chance possible to get those dollars," CEO Terry Gilliland said at the HLTH Conference last month. "We literally are rural health care."
2. Top drug regulator resigns, claims retaliation

George Tidmarsh, the FDA's top drug regulator, abruptly resigned yesterday after federal officials began reviewing "serious concerns about his personal conduct," AP reports.
- In an interview with The New York Times, Tidmarsh claimed a "toxic environment" at the FDA and said the review was retaliation for warnings about politics trumping science in the drug review program.
Tidmarsh's ouster is the latest in a string of leadership changes at the agency, which has been rocked for months by firings, departures and controversial decisions on vaccines, fluoride and other products.
- Tidmarsh, an M.D., was director of the Center for Drug Evaluation and Research, the FDA's largest division, which has lost 1,000+ staffers over the past year to layoffs or resignations.
Tidmarsh was placed on leave Friday, HHS press secretary Emily Hilliard said. Tidmarsh then resigned yesterday.
- A drugmaker connected to one of Tidmarsh's former business associates, Aurinia Pharmaceuticals, said in a lawsuit that Tidmarsh used his FDA position to pursue a "longstanding personal vendetta" against a company executive.
Tidmarsh founded and led a series of pharmaceutical companies over several decades in California's pharmaceutical and biotech industries.
3. FDA plans streamlined gene-editing approach
The FDA is planning to unveil a new approach to approving gene-editing treatments, according to a top official.
Why it matters: The new approach could speed approval of gene-editing therapies that have raised hopes of finding cures to rare diseases through personalized treatments.
Driving the news: Vinay Prasad, a top FDA official, said in an interview with Bloomberg News that the agency will publish the new approach in a paper in early November.
- The traditional approach of conducting separate studies for each new treatment has faced challenges in the new era of gene-editing treatments that can be targeted at a single person.
- Prasad promised to be "extremely flexible," telling Bloomberg that "the moment we publish our paper, the investment in this space will flow."
The big picture: The impending move from the FDA comes after Children's Hospital of Philadelphia announced earlier this year that a child had become the first patient to be successfully treated with a customized gene-editing therapy using technology known as CRISPR.
Our thought bubble: Gene therapies can cost millions of dollars, so how to pay for them has become an urgent question in Washington.
- Sen. Bill Cassidy (R-La.) in particular has solicited ideas for new payment models.
- The financial strain could increase as these therapies become more common, though less time-consuming FDA approval could help lower costs.
4. Medicare finalizes cuts for specialty services
The Trump administration on Friday finalized a controversial plan to reevaluate how Medicare calculates doctor payments that will result in lower rates for specialty services.
Why it matters: Medicare will implement a 2.5% cut next year to payments for services like radiology and gastroenterology that are based on more than time spent delivering the service.
Catch up quick: The rule is based on the CMS argument that these procedures and tasks are being done more efficiently than the old payment rate reflects.
- Meanwhile, primary care doctors and other providers will generally see an increase in their Medicare payment next year.
- The rule moves Medicare away from setting billing code reimbursement based on surveys by an American Medical Association-affiliated committee that ask providers about the time, intensity and expense of different medical services.
What they're saying: The change is an "important step to address methods that have long diverted funding away from the whole-person, relationship-based primary care Americans need," Ann Greiner, CEO of the Primary Care Collaborative, said in a statement.
The other side: Specialists say the policy devalues their work and could result in worse care for patients.
- "These reductions to physician work ignore the realities of modern medicine, including rising patient complexity and evolving technologies that demand more from physicians, not less," Qihui Zhai, president of the College of American Pathologists, said in a statement.
5. Charted: Menopause legislation by state
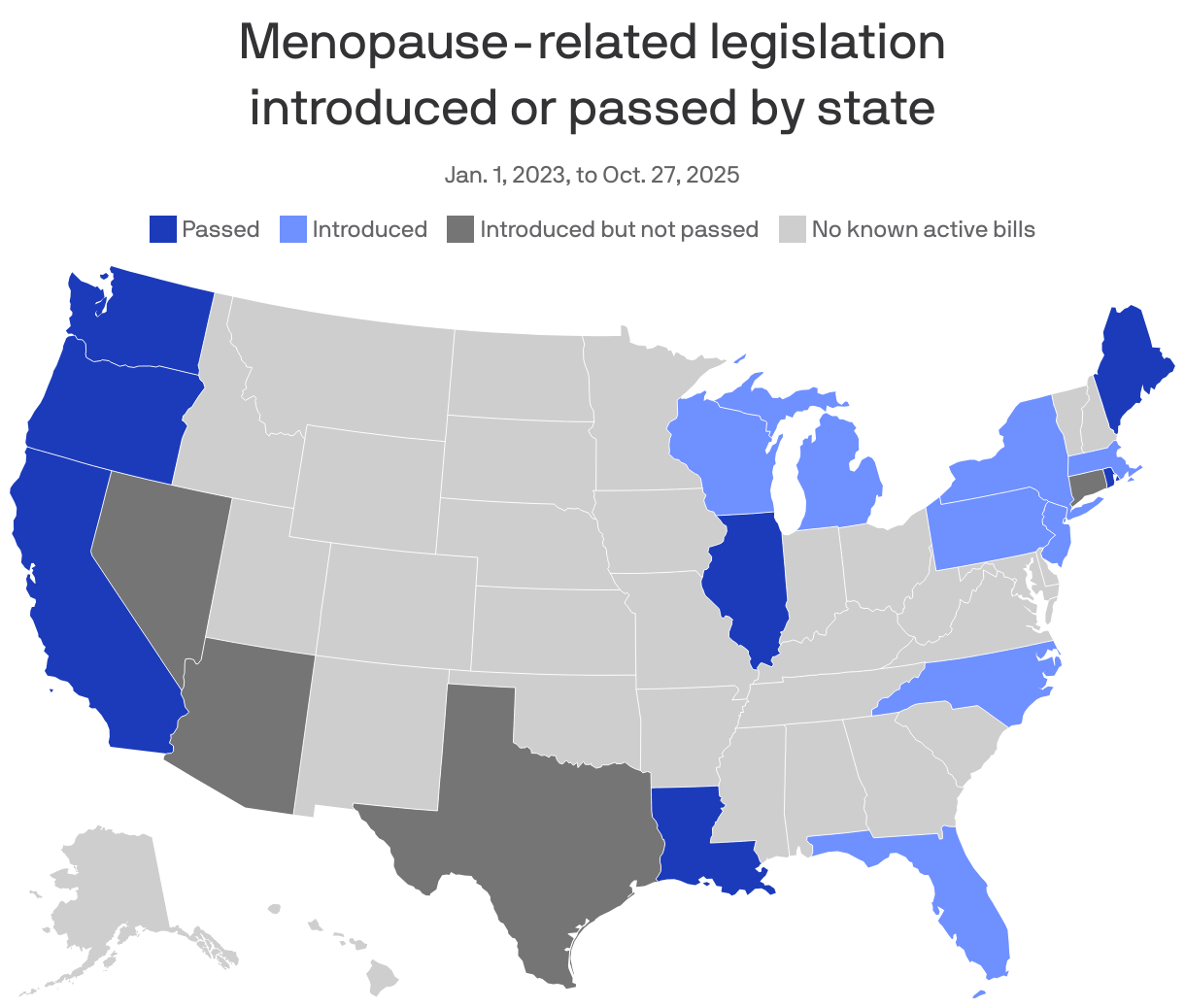
Seven states have passed legislation aimed at promoting menopause care, and at least 36 bills have been introduced in 18 states in a sign of how the condition is becoming more of a policy and health care priority.
Why it matters: Changes like mandates for insurance coverage of treatments in Louisiana and Illinois could not only make treatments more affordable, but bolster workplace support and increase medical training.
- 2025 is already "a watershed year — and was only just the start. Expect to see even more momentum in 2026," Jennifer Weiss-Wolf, executive director of the Birnbaum Women's Leadership Center, told Axios.
State of play: Millennials, the generation that currently includes the most women in America, are heading into the menopause years.
- Celebrities are launching telehealth companies and saying "women deserve better." Influencers are pushing products, which may or may not have scientific backing.
- Gen X and millennial women also are becoming more vocal about the weight gain, hot flashes, brain fog and other symptoms, posting online about their ordeals and pushing providers for better answers.
6. While you were weekending
😳 Obamacare sticker shock after the subsidies expire will depend on where people live, how heavily their premiums have been subsidized and what their income is. (Politico)
💰 At least 31 states have enacted laws this year intended to lower drug costs, and nearly two-thirds are aimed at pharmacy benefit managers. (Stateline)
💊 The Health Resources and Services Administration has approved nine drugs to test a new rebate model for the 340B drug discount program. (The Pharma Letter)
Thanks for reading Axios Vitals, and to managing editor David Nather and copy editor Matt Piper. Please ask your friends and colleagues to sign up.
Sign up for Axios Vitals