Axios Vitals

February 18, 2026
Hello Wednesday! Today's newsletter is 963 words, a 3.5-minute read.
1 big thing: Trump officials seek peace with pharma
Senior Trump administration health officials sought to smooth over simmering tensions with the drug industry at an industry forum on Tuesday — but with limited success.
Why it matters: Drug companies have been frustrated navigating President Trump's pricing initiatives and a turbulent FDA that's drawn criticism for inconsistent standards and anti-vaccine rhetoric.
Driving the news: CMS administrator Mehmet Oz and FDA commissioner Marty Makary both appeared at the PhRMA Forum, a slickly produced annual event at a Washington concert venue where the industry touts its role in finding new cures.
- Oz sought to present Trump's "most-favored-nation" effort — which would tie U.S. drug prices to what's paid in other developed countries — as accommodating to manufacturers.
- "I don't want to hurt innovation," he said. He noted that much of the initiative applies to Medicaid, where "prices are lower anyway, so it's less hurtful; the sector did OK, we believe."
- He said the administration is seeking to get Congress to write most-favored-nation agreements into law because once contracts expire, "if it's not done right, a future administration will take more drastic, draconian steps in ways that would hurt this industry."
Reality check: The message didn't win over the big industry trade group PhRMA.
- The group's CEO, Steve Ubl, told reporters afterward that it unequivocally opposes Congress codifying most-favored-nation pricing, seeing it as "price controls" that will harm innovation.
- He also offered some more measured concern about turmoil at the FDA, noting the importance of a "stable, predictable" agency.
- "Nothing chills investment like uncertainty," he added.
What they're saying: Makary sought to soften some of the edges on the agency's actions on vaccines, including the recent high-profile refusal to review Moderna's mRNA flu vaccine, which sent shock waves through the industry.
2. Cassidy offers Rx for overhauling FDA
Senate health committee chair Bill Cassidy on Tuesday laid out a plan to overhaul the FDA that calls for more predictability in the drug review process and for speeding up development of drugs for rare diseases.
Why it matters: The recommendations aren't likely to become law but could keep the congressional spotlight on agency decision-making that many lawmakers believe has been inconsistent.
What's inside: Cassidy (R-La.) called for addressing instances where companies face what they term a "reviewer lottery," where critical questions hinge on the approach of a small number of individuals at FDA.
- He said one way to improve predictability is by adapting principles in a 1997 law that required FDA to take a "least burdensome" approach to regulating medical devices.
- Cassidy also raised concern about the use of "clinical holds" that freeze studies on drugs for rare diseases, and questioned if Congress should act to increase competition from generic drugs and shorten approval of biosimilars.
The plan didn't reference the widely prescribed abortion drug mifepristone. Cassidy previously called on FDA to complete a safety review of the pills and to reinstate in-person dispensing requirements.
3. Low CMS rates blunt digital health hopes
Digital health insiders are underwhelmed by the reimbursement rates CMS just set for a tech-focused pilot program that pays Medicare providers a fixed amount to manage patients' chronic issues.
Why it matters: The industry hoped for a major health tech boost from the ACCESS program, but the $7 per-member, per-month rate is a far cry from the $30-$50 figure industry experts anticipated, Erin Brodwin wrote first on Pro.
What they're saying: The figures "are so low that it will require some real breakthroughs in technology-enabled care to make the math work," says Venrock partner Bob Kocher.
- "This is not enough money to support clinicians engaging in care," Kocher adds.
How it works: CMS' initial reimbursement figures range from roughly $180-$360 per year, depending on the specialty.
- The agency then withholds half until outcomes are proven at the end of a 12-month care period.
Case in point: For musculoskeletal conditions, for example, the agency will reimburse $180 per year.
- With $90 withheld, that leads to roughly $7.50 in monthly cash flow per patient.
The other side: The model is better suited for companies making intensive use of AI — letting them operate with less staff and on slimmer margins, several industry experts say.
If you need smart, quick intel on health tech dealmaking for your job, get AxiosPro.
4. Charted: Abortion clinic access by state
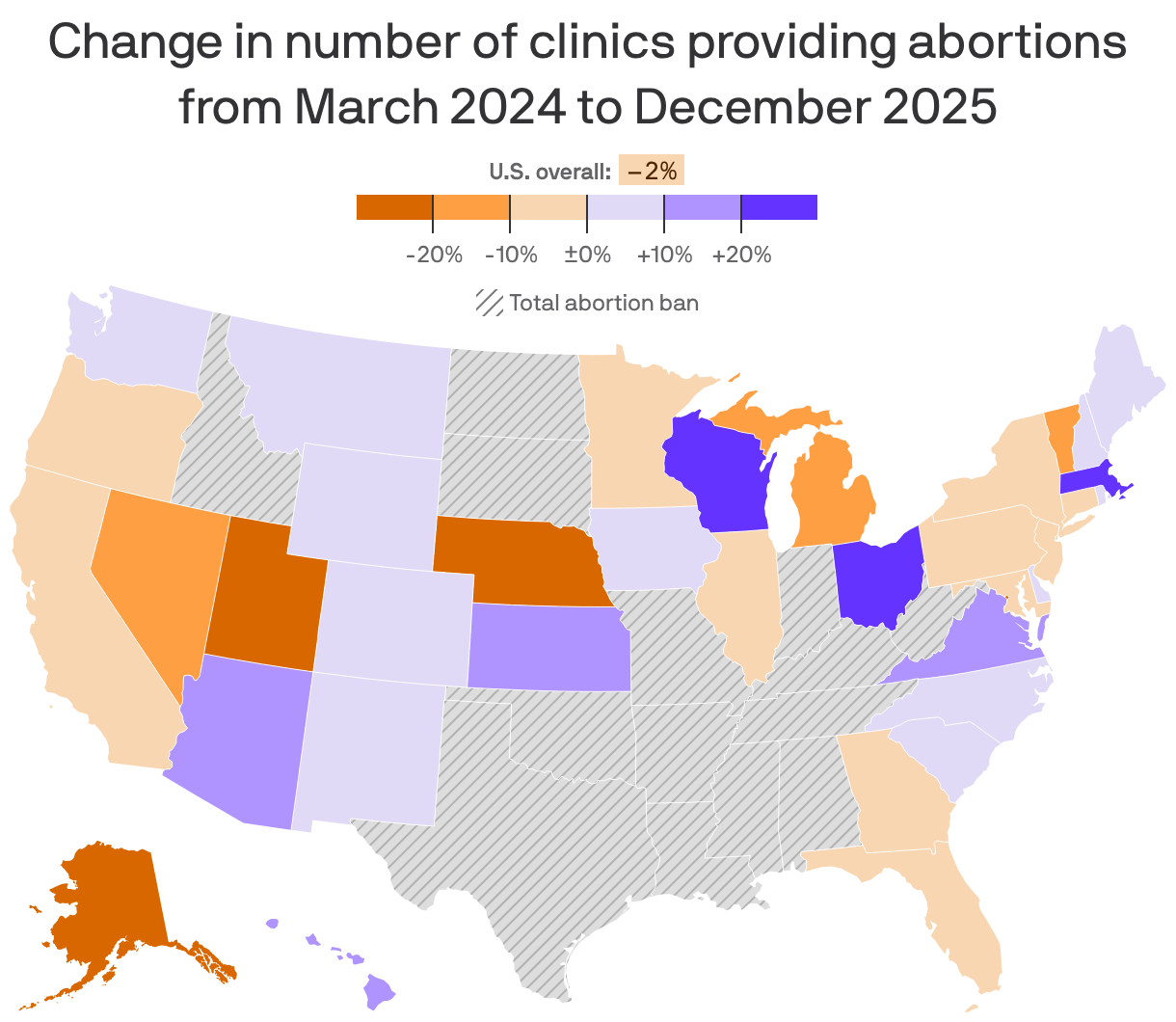
Federal funding freezes and new abortion restrictions since March 2024 likely factored in the closing of abortion clinics and limited access to the procedure in other health settings, the Guttmacher Institute reports in an analysis out today.
Why it matters: Physical clinics still provide about 80% of abortions in states without abortion bans, even as telehealth-prescribed medication abortion becomes more prevalent.
What they found: The Trump administration's withholding of Title X funds from reproductive health networks such as Planned Parenthood last year likely contributed to the closure of three clinics in Michigan.
- Florida's ban on most abortions after six weeks that took effect in May 2024 similarly factored in the closing of four clinics in the state.
- But Missouri saw the opening of three clinics following voters' reversal of the state's post-Roe abortion ban in November 2024.
Online-only clinics have become a critical new source of care, Guttmacher notes, even though brick-and-mortar clinics are usually the only option for those who need or prefer a procedural abortion.
5. Catch up quick
⚖ Bayer struck an agreement to pay $7.25 billion to settle tens of thousands of lawsuits that claimed the weedkiller Roundup caused non-Hodgkin's lymphoma. (NYT)
💉 California is preparing a lawsuit challenging recent federal changes to U.S. childhood vaccine recommendations made by the Trump administration, the state's attorney general said. (Reuters)
🏥 Trump required hospitals to post their prices. So far, there's no evidence that patients use the information. (KFF Health News)
Thanks for reading Axios Vitals, and to editors Adriel Bettelheim and David Nather and copy editor Amy Stern. Please ask your friends and colleagues to sign up.
Sign up for Axios Vitals






