Infant formula safety: FDA doesn't require botulism test
Add Axios as your preferred source to
see more of our stories on Google.
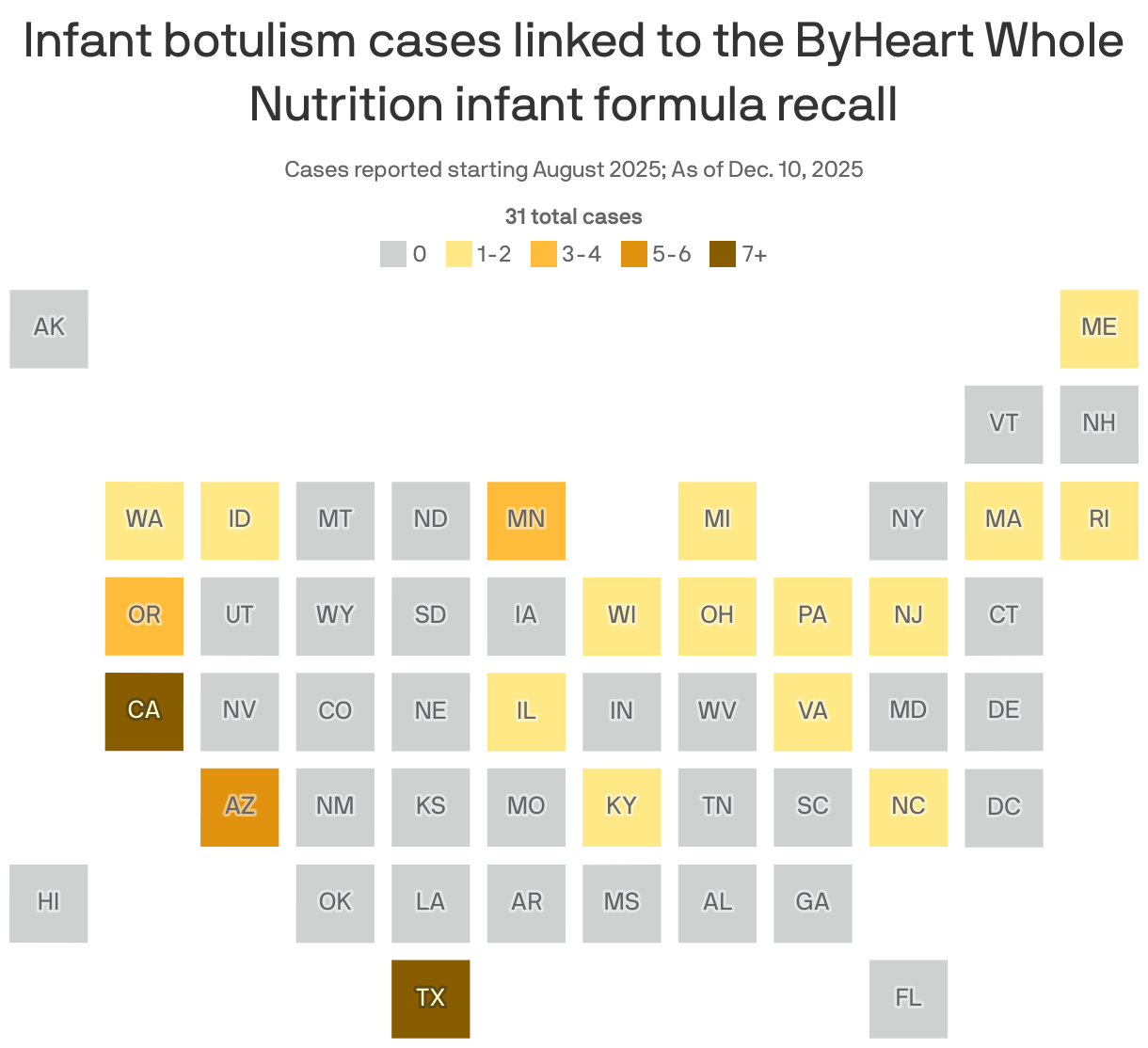
As of Dec. 10, 51 infants have been hospitalized for suspected or confirmed botulism in an outbreak linked to ByHeart baby formula, the FDA says.
Why it matters: Though botulism in infants is rare, it's become a new concern for parents of formula-fed babies.
- The FDA doesn't require formula companies to screen products for the bacteria that causes botulism.
What's happening: Infant botulism happens when a baby swallows Clostridium botulinum (also called C-bot) spores. Then, a toxin is produced as the bacteria grows in the gut. It can be fatal if left untreated.
- Botulism can first present as constipation, then difficulty feeding, a weak cry and lack of head control.
- Symptoms can take several weeks to develop and can be treated with the drug BabyBIG.
- Honey is the best-known carrier of the spores that cause infant botulism, which is why honey is not recommended for babies.
The latest: No deaths have been reported to date.
- The CDC had been counting botulism cases from Aug. 1 of this year onward, but it recently expanded the outbreak definition to include years before that: Ten additional cases, with documented exposure to ByHeart Formula, occurred from Dec. 2023 through July 2025.
What they're saying: When ByHeart announced it would begin screening for C-bot after the botulism outbreak, "My reaction was, 'Why weren't you already doing that test?'" Frank Yiannas, a former deputy commissioner for food policy at the FDA, tells Axios. "The leading edge science-based organizations that have been producing powdered infant formulas for years, they're aware of this, and they already screened for this."
- "Until now, this bacterium was not among the pathogens routinely tested for across the industry, despite thousands of safety tests conducted by all manufacturers," ByHeart said in a statement to Axios. "We are committed to using this moment to make our ByHeart processes and the industry at-large safer for families."
State of play: Sulfite-reducing clostridia (SRC) screening tests can flag the presence of a high level of spores and increased risk for botulism. The tests are not FDA required, but they're also "not novel or new," Yiannas says.
- Health and Human Services Secretary Robert F. Kennedy's Operation Stork Speed, a review of the infant formula food supply, has promised to screen for more contaminants in infant formula, but it hasn't said how or whether C-bot screenings will be part of the requirements.
What we're watching: After C-bot was identified in unopened ByHeart formula, an investigation by federal and state health officials seeks to pinpoint the cause of the infant botulism outbreak and determine whether it was tied to a specific ingredient or another part of the ByHeart supply chain.
Editor's note: This story was originally published on Nov. 26 and has been updated with new case numbers.
