The ascent of a $225,000 heart therapy
Add Axios as your preferred source to
see more of our stories on Google.
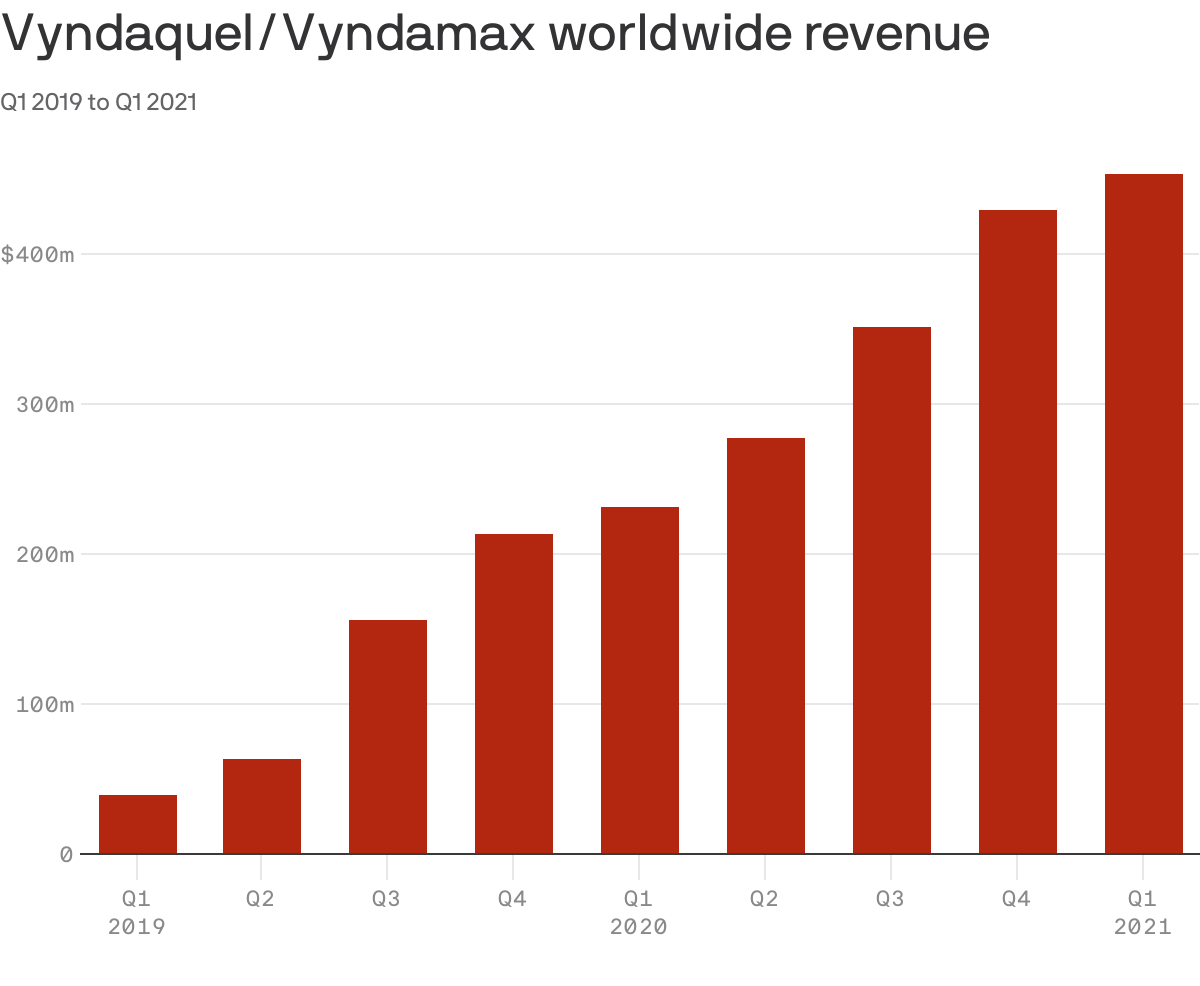
Heart medications Vyndaqel and Vyndamax generated $1.3 billion of global revenue last year for Pfizer and already brought in $453 million in the first quarter of this year.
Why it matters: The blockbuster drugs could grow significantly more if Pfizer wins its pending lawsuit and is allowed to pay the out-of-pocket expenses of Medicare patients who are prescribed the $225,000-a-year treatment.
Between the lines: The drugs, which both have the active ingredient tafamidis, have a complex history.
- Pfizer didn't develop the drug. It acquired the drug's developer, FoldRx Pharmaceuticals, in 2010 for an undisclosed amount.
- Pfizer then got the drug through a pivotal phase 3 trial and won FDA approval in 2019 after the FDA originally rejected the drug in 2012.
- The National Institutes of Health funded the basic research that led to tafamidis.
- Researchers say an annual price of $16,500, or a 93% discount, would match the drug's benefits. The United Kingdom won't cover tafamidis due in large part to the price. Pfizer does not disclose the drug's net price after rebates.
A bit of foreshadowing: "We're exploring ... a number of ways that we can help lower copay costs for Medicare patients," Pfizer executive Angela Hwang told investors in 2019, after Pfizer got FDA approval but before it filed its lawsuit.
