Oct 31, 2020 - Health
Exclusive: Fully at-home rapid COVID test to move forward
Add Axios as your preferred source to
see more of our stories on Google.
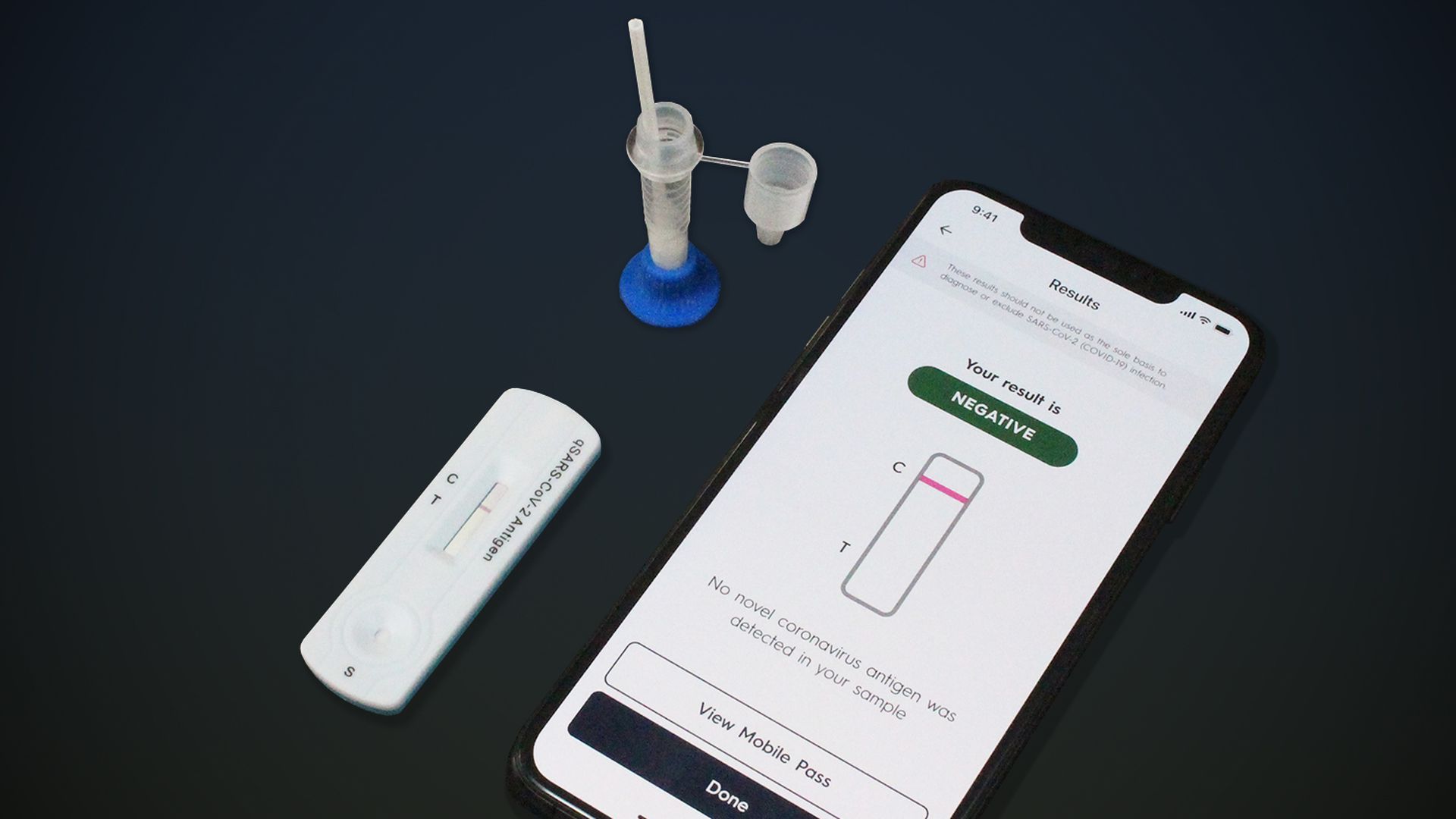
Gauss/Cellex rapid at-home COVID-19 test. Photo: Courtesy of Gauss
Add Axios as your preferred source to
see more of our stories on Google.

Gauss/Cellex rapid at-home COVID-19 test. Photo: Courtesy of Gauss