Apr 16, 2018 - Health
Ebola vaccine shows long-term protection over 2 years
Add Axios as your preferred source to
see more of our stories on Google.
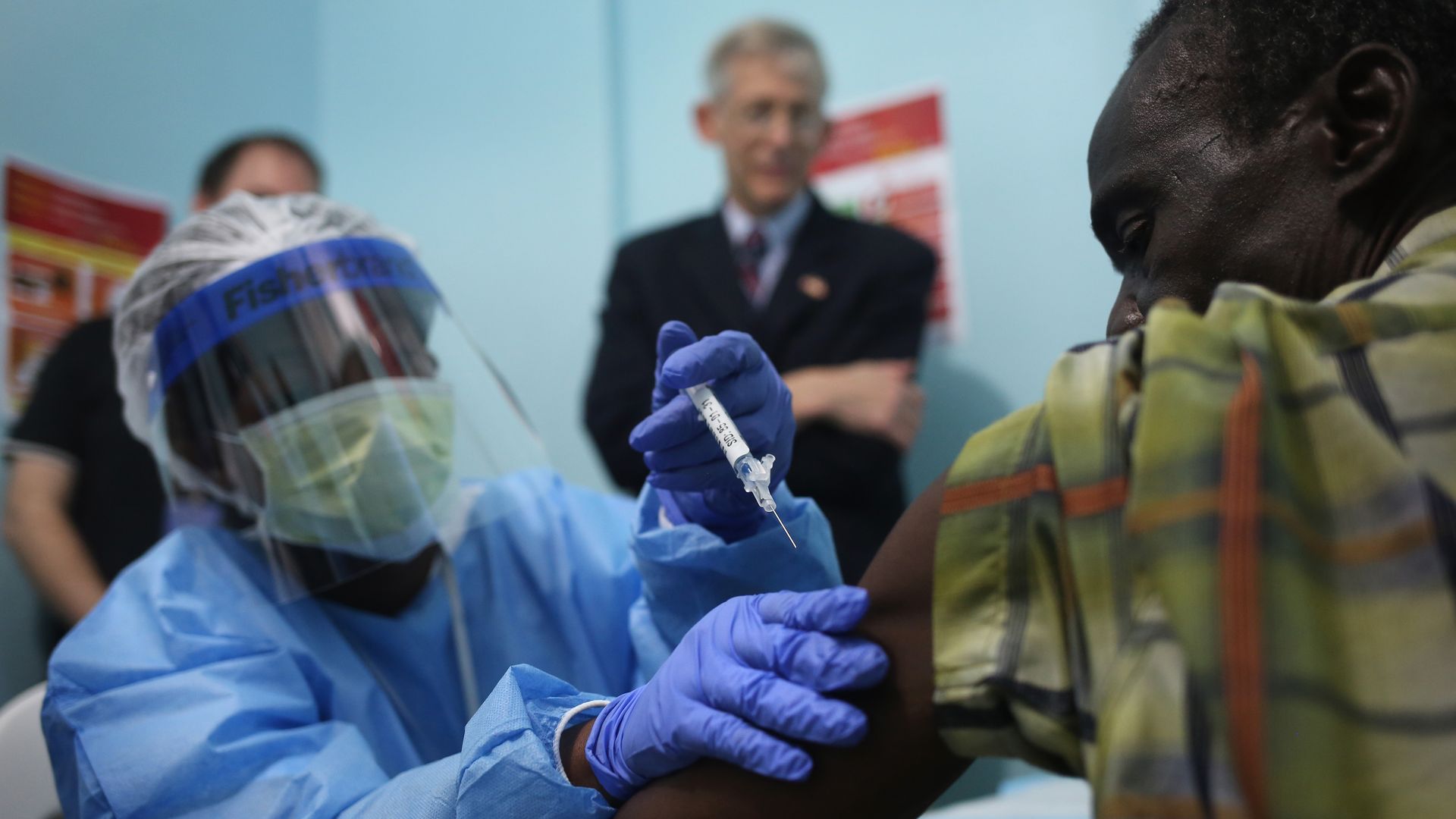
A nurse administers an Ebola vaccine in Monrovia, Liberia, as part of a 2015 clinical research study. Photo: John Moore/Getty Images
