Axios Vitals

March 24, 2026
Good morning. Today's newsletter is 1,100 words, a 4-minute read.
⚡️ Join Axios on Thursday, March 26, at 8am in Washington, D.C., for an event exploring rising health care costs, featuring Sen. Lisa Blunt Rochester (D-Del.), Rep. Adrian Smith (R-Neb.) and more. RSVP here.
1 big thing: Trump visa freeze sidelines doctors
The Trump administration's suspension of certain immigrants' work authorization renewals is sidelining possibly thousands of foreign-born doctors, some of the affected physicians tell Axios.
Why it matters: The policy could worsen access to care in a health system already facing physician shortages.
- About a quarter of the U.S. physician workforce are immigrants. Rural communities especially depend on immigrant doctors to fill workforce gaps.
State of play: The Citizenship and Immigration Services this winter started freezing the processing of immigration benefits like work authorization extensions for people already in the U.S. from 39 countries.
- The change has affected work authorization renewals, green card processing and naturalization.
Where it stands: Visa holders have a grace period allowing them to continue working in the U.S. after applying for a renewal. For the common H-1B visa, that period is 240 days.
- But the freeze has scrambled arrangements, forcing doctors to take unpaid absences from their jobs. They can legally stay in the U.S. if their employer continues to sponsor them.
- Immigrant doctors caught in the squeeze now either have to return to their home countries, immigrate elsewhere or stay in the U.S. unable to work.
What they're saying: "Even with us, there is [a] severe shortage," one Ohio-based doctor told Axios. "Can you imagine any physician loss, how it will impact the society here?"
- He and others interviewed were granted anonymity to discuss their immigration status due to fear of retaliation.
Patients already are feeling the fallout. The Ohio doctor said his clinic patients had to be rescheduled to other doctors when his work authorization ran out in February.
- Those patients may have to wait months for a new appointment, he said. Other doctors picked up his on-call shifts, stretching their own workloads.
- The American Medical Association wrote to the Department of Homeland Security that one doctor estimated their work suspension left more than 900 patients without sufficient care.
- "It's not just an immigration issue," said a Michigan doctor whose work authorization runs out in July. "It's a patient care issue that's hiding in plain sight."
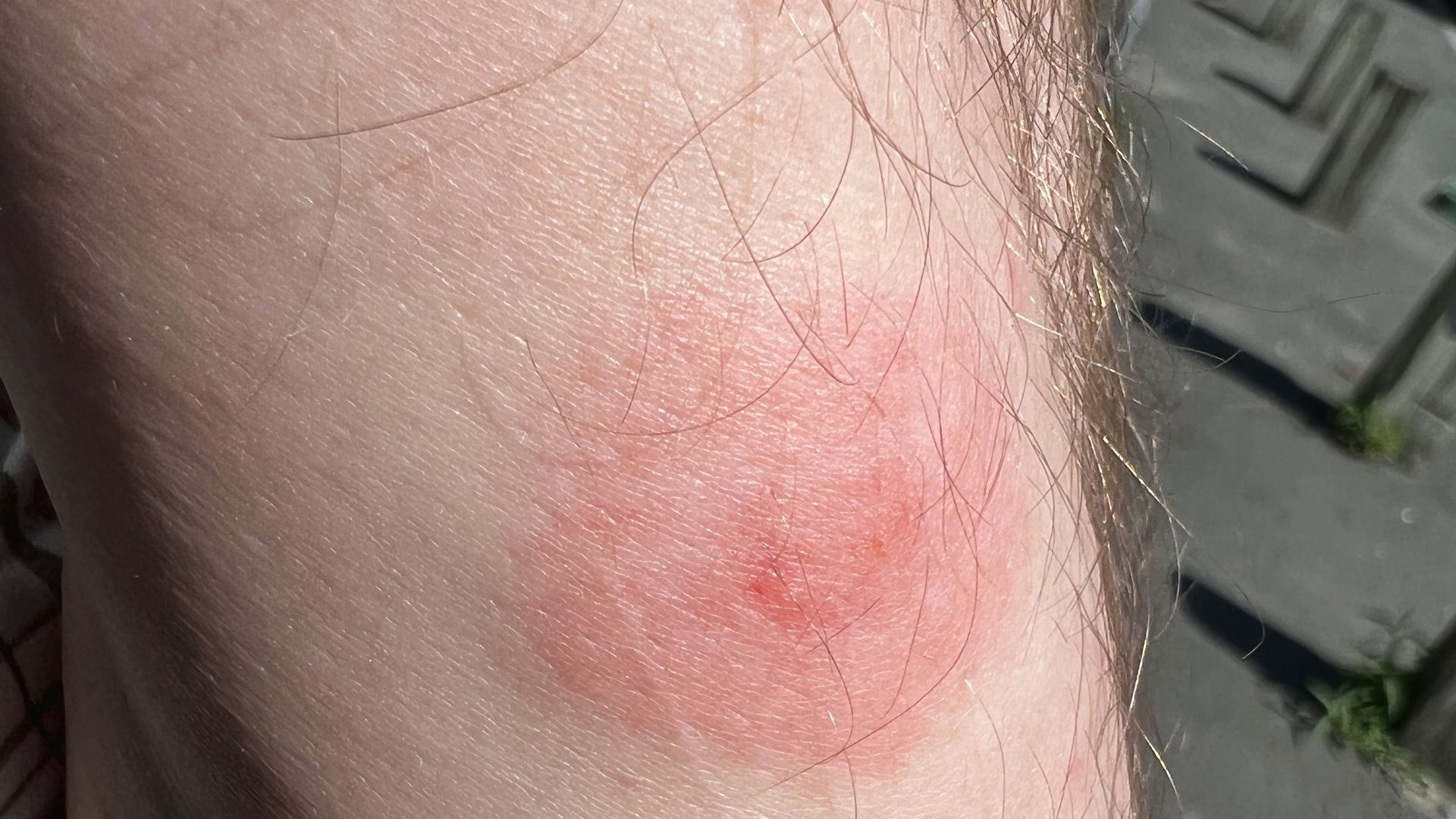
2. Lyme disease vaccine effort to move forward
A Pfizer-led effort to develop the first new Lyme disease vaccine in more than two decades is moving forward despite missing its statistical target in a closely watched clinical trial.
Why it matters: There are no approved human vaccines for Lyme disease, which affects nearly half a million people in the U.S. each year.
Driving the news: Pfizer and its partner Valneva said yesterday their experimental shot showed more than 70% efficacy in preventing the tick-borne illness in individuals age 5 and above.
- The companies said they missed a predetermined target because there were fewer-than-expected cases studied over the course of the trial.
- Despite that, the partners said the results strengthen their confidence in the vaccine, and that Pfizer is planning submissions for regulatory approval.
The intrigue: The prospects at the FDA are unclear. Health Secretary Robert F. Kennedy Jr. signaled a new focus on Lyme disease when he convened a roundtable of patients, clinicians and researchers in December.
- But Trump administration regulators have also called for tighter government vaccine oversight, including stricter guidelines for new vaccines.
- If the shot is approved, adoption is likely to be limited to "a small percentage of people who are motivated to seek out a four-dose vaccine which showed a 73% reduction in the rate of confirmed Lyme disease cases," Leerink analyst David Risinger wrote in a note.
There hasn't been a commercially available Lyme disease shot since a vaccine called LYMERix was pulled from the market in 2002 due to low consumer demand tied to reports of adverse events.
- The public has had to rely on antibiotics like doxycycline after a tick bite.
3. Using several AI health tools could pose risks
Hundreds of AI tools for health care — from transcription and imaging to diagnostics — tout accuracy rates above 90%, but most are tested only in isolation, Erin Brodwin wrote first on Pro.
Why it matters: Those tools become less reliable when used together, an analysis by Korean AI scientist Kwansub Yun suggests.
Inside the room: Yun and health consultant Claire Hast ran an example scenario in which a patient had a physical transcribed by AI, received a mammogram using AI-assisted imaging and got a diagnosis with help from an AI tool.
Stunning stat: While each tool individually had a reported accuracy rating of more than 85%, the system as a whole had a reliability score of just 74%.
Between the lines: In practical terms, if erroneous data from one AI tool is fed into another, the secondary tool has no way to flag the unreliable inputs, says Yun.
- "The result looks authoritative, but the chain that produced it was never measured end to end."
Friction point: That's particularly troubling given that the standard regulatory procedure for evaluating the tools involves standalone model performance testing, Hast and Yun say.
If you need smart, quick intel on health tech dealmaking for your job, get Axios Pro.
4. Novartis boosts investments in China
Swiss drug giant Novartis is the latest big pharmaceutical company announcing plans to bolster its research and production in China.
Why it matters: Global life sciences companies are increasingly clustering in life sciences hubs in China and also licensing early-stage experimental drugs made by Chinese companies.
- That's threatening the United States' long-standing dominance in drug-making.
Driving the news: Novartis said it's investing the equivalent of $480 million in a Beijing-area manufacturing plant and a Shanghai research campus that's home to its China headquarters.
- The Beijing plant has a maximum annual production capacity of 3 billion tablets and capsules and 550 million boxes of packaging, the company wrote in a WeChat post.
- The company's areas of focus include developing targeted radioactive cancer treatments.
Novartis also is investing $23 billion to build or expand 10 U.S. facilities — a commitment President Trump said was the result of his tariff strategy.
5. Catch up quick
💵 The administration approved a plan submitted by Minnesota to combat Medicaid fraud, possibly staving off billions of dollars in funding cuts. (Politico)
🗳️ Polls show that when voters say that affordability is their biggest concern, for many, they're specifically talking about health care. (NYT)
📦 The nationwide shortage of stimulant medications used to treat ADHD may be rooted less in overprescribing than in global supply chain disruptions. (CIDRAP)
Thanks for reading Axios Vitals, and to editors Adriel Bettelheim and David Nather and copy editor Matt Piper. Please ask your friends and colleagues to sign up.
Sign up for Axios Vitals