Axios Vitals

February 09, 2026
Welcome back, Vitals gang. Today's newsletter is 1,089 words, a 4-minute read.
1 big thing: Limits on care denials in Medicare stall
Long-running efforts to limit health plans' ability to deny or delay physician-ordered care are on hold again, despite mounting congressional frustration with insurers.
Why it matters: By leaving a measure addressing so-called prior authorization reviews out of the bipartisan health package last week, lawmakers may have lost the chance to address a major source of aggravation for patients and doctors before the midterms.
Driving the news: Efforts to streamline the prior authorization process in Medicare Advantage have come close in recent sessions of Congress before stalling over cost concerns.
- This year, one such measure — which would take steps to require insurers to cut down the time to make decisions and adopt electronic systems to smooth the process — was left out of the health package that was folded into the latest government funding deal.
- "It's a missed opportunity, but it's not the last opportunity," said Charlene MacDonald, CEO of the Federation of American Hospitals. "That certainty that seniors are going to get the care that they deserved in a timely manner is still important."
Between the lines: Republicans and President Trump have increased their attacks on health insurers, including bringing in executives for a hearing last month that showcased bipartisan concerns over prior authorization.
- "Republicans today sound a lot like Democrats have sounded for a long time, saying there's way too much profit, they're taking too much money out of the system," said Rep. Ami Bera (D-Calif.), who's pushing for changes.
- But it remains to be seen if the rhetoric translates into any action.
There were nearly 53 million prior authorization requests in Medicare Advantage in 2024, per KFF, with about 8% fully or partially denied. Government auditors have also found high rates of denials in Medicaid-managed care plans.
- The Biden administration tried to address the issue through regulations, but Congress split in 2024 on whether to simply codify those rules or add changes backed by Democrats that addressed the use of AI to deny medically necessary care.
2. Cheaper alternatives for some TrumpRx drugs
More than half of the drugs listed on the new TrumpRx website have or will soon have a cheaper generic version on the market that can be purchased through other direct-to-consumer sites.
Why it matters: Patients could save more buying nonbrand copycat drugs, even if they don't have insurance and pay cash, drug pricing experts say.
Case in point: Pristiq, an antidepressant made by Pfizer, is available with a TrumpRx coupon for about $200 for a 30-day supply — a more than 50% discount from the list price.
- But a month's worth of a generic version with the same dosage is available on GoodRx for less than $30, and on Mark Cuban's CostPlusDrugs for just $16.65.
Where it stands: 20 of the 43 drugs listed on TrumpRx as of the website's launch have generic alternatives, according to a tally by Anna Kaltenboeck, a drug pricing expert and president of Verdant Research.
- Another six have components that are available as generics or compounded products or that have tentative approvals, meaning consumers will have access to the lower-cost products in the near future.
- "To the extent that a patient was buying these without insurance, I don't see this being a benefit," Kaltenboeck said.
The intrigue: TrumpRx could help "funnel people to higher price-products and [take] money from them that they don't need to be spending," said Sarah Karlin-Smith, research director for the Access to Medicines program at Public Citizen.
A White House spokesperson said the site's value is in providing the lowest-cost option for branded products.
3. Charted: Trust in CDC after vaccine changes
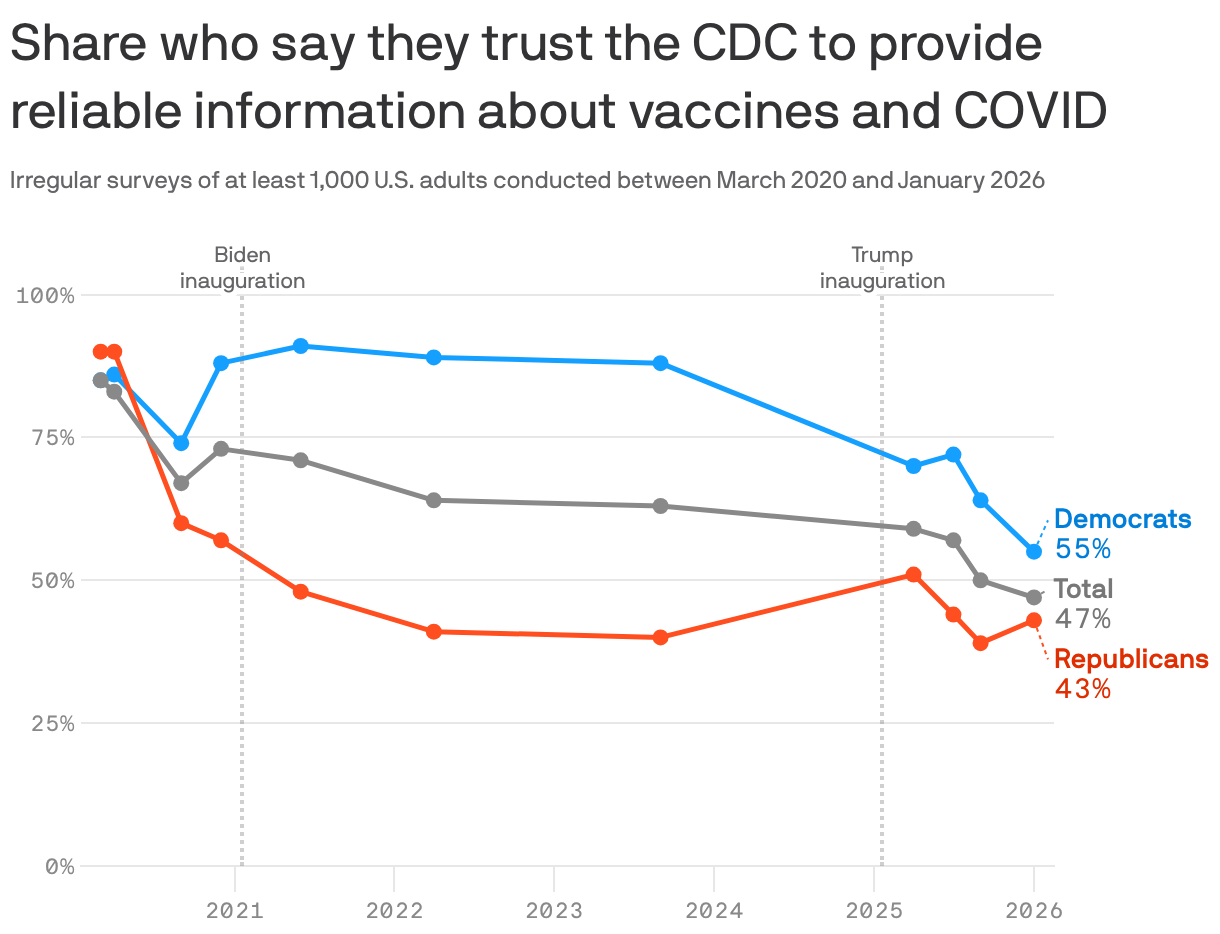
The public's trust in the CDC continues to decline, with a pronounced drop among Democrats since the Trump administration's changes to the childhood vaccine schedule, according to the latest KFF tracking poll.
- While trust among Democrats declined by nine percentage points since September, about 4 in 10 Republicans (43%) and half of independents (46%) say they trust the the agency for reliable information about vaccines.
- At least 8 in 10 U.S. adults across across party lines are confident in the safety of measles, mumps, and rubella (MMR) and polio vaccines, which the CDC continues to recommend as routine.
- Majorities are also confident in the safety of two of the vaccines that are no longer universally recommended for kids: hepatitis B (70%) and flu (65%), though views are divided along partisan lines.
- Fewer than half of Republicans and self-described supporters of the "Make America Healthy Again" movement say they trust federal health agencies to make recommendations about vaccine schedules, per KFF.
4. Hims & Hers backs off compounded GLP-1 pill
Hims & Hers retreated from plans to sell a cheaper copycat version of Novo Nordisk's Wegovy pill after HHS asked for an investigation of whether the telehealth brand violated the Food, Drug and Cosmetic Act.
Why it matters: The developments — which also included threatened litigation from Novo Nordisk — underscored continued tension over compounded GLP-1 weight loss drugs and when companies can make customized versions tailored to patients' needs.
- Hims & Hers has relied on sales of compounded versions of GLP-1 injections to fuel its growth. But in September, the FDA warned the company about what regulators said were misleading claims implying its products are the same as FDA-approved drugs.
Driving the news: Last week, Hims & Hers advertised a version of the Wegovy pill for $49 for the first month and $99 a month thereafter — an introductory price roughly $100 less than Novo Nordisk's.
- HHS general counsel Mike Stuart wrote on X Friday that the company had been referred to DOJ for potential violations of the Food, Drug and Cosmetic Act following a review.
- FDA commissioner Marty Makary also warned his agency would take "decisive steps" to restrict GLP-1 active ingredients intended for use in non-FDA-approved compounded drugs that are mass-marketed by companies including Hims & Hers.
- Hims & Hers said in a statement on Saturday that it decided to stop offering access to the treatment after "constructive conversations with stakeholders across the industry."
5. While you were weekending
⚕️ Nearly five years after Texas criminalized abortion, its state medical regulator is issuing training on when a pregnancy can be terminated to protect a patient's life. (ProPublica)
💊 HHS formally scrapped a drug pricing pilot that would have allowed manufacturers to give post-sales rebates instead of upfront discounts. (Healthcare Dive)
🗳️ Health costs are stoking voter anxiety and powering Democratic campaigns, with spots focusing on struggling hospitals and spiking insurance premiums. (AP)
Thanks for reading Axios Vitals, and to editors Adriel Bettelheim and David Nather and copy editor Matt Piper. Please ask your friends and colleagues to sign up.
Sign up for Axios Vitals








